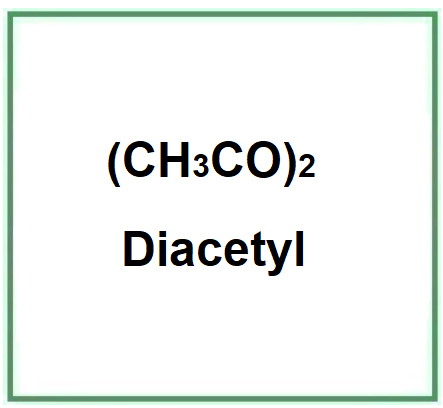
Diacetile (2,3 butanedione) è un composto chimico organico, un dichetone vicinale volatile.
Il nome definisce la struttura della molecola
- "Di" indica la presenza di due gruppi funzionali nella molecola.
- "Acetyl" si riferisce al gruppo funzionale acetile, che è derivato dall'acido acetico.
Descrizione delle materie prime utilizzate nella produzione
- Cultura batterica - Ad esempio, il batterio Lactobacillus utilizzato nella fermentazione.
- Substrati zuccherini - Come glucosio o maltosio.
Descrizione delle materie prime utilizzate nella produzione
- Preparazione del substrato - I substrati zuccherini sono preparati e solubilizzati in acqua.
- Innoculazione - La cultura batterica viene introdotta nel substrato zuccherino.
- Fermentazione - La cultura batterica fermenta gli zuccheri e produce diacetil come un sottoprodotto. Questo avviene in condizioni anaerobiche controllate.
- Estrazione - Dopo un periodo di tempo determinato, il diacetile è estratto dalla soluzione di fermentazione.
- Purificazione - Il diacetile viene purificato per rimuovere impurità.
Si presenta in forma di liquido incolore.

A cosa serve e dove si usa
Cosmetica
Profumo. A differenza della fragranza che può contenere anche odori leggermente meno gradevoli o caratteristici, la dizione profumo indica soltanto le profumazioni molto gradevoli. Utilizzato per profumi e materie prime aromatiche.
Alimentazione
Diacetile è un sottoprodotto della fermentazione in bevande alcoliche ed è aggiunto come aromatizzante sintetico negli alimenti per conferire un sapore burroso. Presente naturalmente in alcune bevande alcoliche come birra e vino a causa della fermentazione.
Sigarette elettroniche
Diacetile viene inserito come agente aromatizzante sintetico.
Sicurezza
Diacetile ha dimostrato, in numerosi studi condotti negli ultimi anni, una tossicità che ha preoccupato gli enti di regolamentazione (1) e la sua esposizione ad alte concentrazioni di vapore può provocare compromissioni a lungo termine nella funzione polmonare (2) nonché un'interruzione dell'omeostasi del ferro (3) e cambiamenti metabolici nella struttura delle proteine (4).
L'esposizione prolungata e significativa al diacetyl, specialmente in contesti industriali, è stata associata a una malattia polmonare chiamata "bronchiolite obliterante" o "polmone da popcorn".
- Molecular Formula C4H6O2
- Molecular Weight 86.09 g/mol
- CAS 431-03-8
- UNII K324J5K4HM
- EC Number 207-069-8
- DTXSID6021583
- Nikkaji J2.586K
- Metabolomics Workbench ID 5418
Synonyms:
- 2,3-Butanedione
- Biacetyl
- Diacetyl
- Diketobutane
- Dimethyldiketone
- Dimethylglyoxal
Bibliografia_____________________________________________________________________
(1) Shibamoto T. Diacetyl: occurrence, analysis, and toxicity. J Agric Food Chem. 2014 May 7;62(18):4048-53. doi: 10.1021/jf500615u. Epub 2014 Apr 25. PMID: 24738917.
Abstract. Diacetyl possesses a butter-like flavor and has been widely used as a flavoring agent. It forms from sugars and lipids via various bacteria and heat treatment in various foods and beverages, such as milk. The toxicity of diacetyl, especially when inhaled, has recently attracted the attention not only of consumers but also of regulatory agencies. Even though accurate quantitative analysis of diacetyl is extremely important in evaluating its possible adverse effects, precise quantitative analysis of diacetyl in foods and beverages, as well as in ambient air, is considerably difficult because it is highly reactive and soluble in water. Among the many analytical methods developed for measuring diacetyl, preparation of 2,3-dimethylquinoxaline followed by gas chromatography has been most commonly used in the analysis of various foods, beverages, and air samples. This mini-review summarizes the formation mechanisms, analytical methods, occurrence, and toxicity of diacetyl.
(2) Brass DM, Palmer SM. Models of toxicity of diacetyl and alternative diones. Toxicology. 2017 Aug 1;388:15-20. doi: 10.1016/j.tox.2017.02.011.
(3) Ghio AJ, Soukup JM, Dailey LA, Roggli VL, Crumbliss AL, Palmer SM. Diacetyl exposure disrupts iron homeostasis in animals and cells. Inhal Toxicol. 2021 May-Jul;33(6-8):268-274. doi: 10.1080/08958378.2021.1989092.
Abstract. Objective: Several mechanisms have been proposed for the biological effect of diacetyl. We tested the postulate that animal and cell exposures to diacetyl are associated with a disruption in iron homeostasis....Results: After exposure of animals to diacetyl, there were airway polypoid lesions which stained positively for both iron and the intracellular storage protein ferritin. Trichrome stain showed a deposition of collagen immediately adjacent to accumulated metal following diacetyl exposure. In in vitro cell exposures, FAC increased non-heme iron concentration but co-incubations of FAC and diacetyl elevated levels to significantly greater values. Levels of ferritin were increased with exposures of BEAS-2B and THP-1 cells to FAC but were similarly greater after co-exposure with FAC and diacetyl. Conclusions: Results of animal and cell studies support a disruption of iron homeostasis by diacetyl. It is proposed that, following internalization, diacetyl complexes intracellular sources of iron. The cell recognizes a loss of its requisite iron to diacetyl and imports greater concentrations of the metal.
(4) Jedlicka LDL, Silva JDC, Balbino AM, Neto GB, Furtado DZS, da Silva HDT, Cavalcanti FBC, van der Heijden KM, Penatti CAA, Bechara EJH, Assunção NA. Effects of Diacetyl Flavoring Exposure in Mice Metabolism. Biomed Res Int. 2018 Jun 28;2018:9875319. doi: 10.1155/2018/9875319.
Abstract. Diacetyl is a flavoring that imparts a buttery flavor to foods, but the use or exposure to diacetyl has been related to some diseases. We investigated the effect of oral intake of diacetyl in male and female C57/Bl mice. We performed a target metabolomics assay using ultraperformance liquid chromatography paired with triple quadrupole mass spectrometry (UPLC-MS/MS) for the determination and quantification of plasmatic metabolites. We observed alterations in metabolites present in the urea and tricarboxylic acid (TCA) cycles. Peroxynitrite plasmatic levels were evaluated by a colorimetric method, final activity of superoxide dismutase (SOD) was evaluated by an enzymatic method, and mouse behavior was evaluated. Majority of the assay showed differences between control and treatment groups, as well as between genders. This may indicate the involvement of sex hormones in the regulation of a normal metabolic profile, and the implication of sex differences in metabolite disease response.
![]() Diacetile
Diacetile