![]() Etidronic acid
Etidronic acid
Rating : 6.5
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
Cons:
Cosmetics Regulation provisions (1)8 pts from FRanier
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.Evaluate | Where is this found? |
| "Descrizione" about Etidronic acid Review Consensus 8 by FRanier (9960 pt) | 2024-Oct-04 16:47 |
| Read the full Tiiip | (Send your comment) |
Etidronic acid (1-hydroxyethylidene-1,1-diphosphonic acid o HEDP) is a phosphonate derived from phosphorus.
The name defines the structure of the molecule:
- Ethyldronic is derived from 'ethane', an alkane that indicates the presence of two carbon atoms in the molecule. The 'Hydron' part is derived from 'hydrogen,' referring to the hydrogen atoms in the molecule.
- Acid. This term refers to a substance that can donate a proton or accept a pair of electrons in reactions.
The synthesis of ethyldronic acid typically involves the reaction of phosphorous acid (H3PO3) with acetic anhydride at elevated temperatures. The synthesis process takes place in several stages:
- Preparation of materials. Phosphorous acid is commercially available. Acetic anhydride or acetyl chloride, which are used as the acetylating agent, are also commercially available.
- Acetylation. Phosphorous acid is reacted with acetic anhydride or acetyl chloride at elevated temperatures and in anhydrous conditions to prevent hydrolysis of the acetylating agent. The reaction can be represented as follows:
2H3PO3 (phosphorous acid) + (CH3CO)2O (acetic anhydride) -> C2H8O7P2 (ethyldronic acid) + CH3COOH (acetic acid)
- Purification. The crude ethyldronic acid product may contain unreacted phosphorous acid and acetic anhydride as well as acetic acid produced as a by-product and therefore must be purified to remove these impurities through various methods, such as crystallization, filtration or extraction.
- Characterization. The final stage of the synthesis process is the characterization of the product to confirm its identity and purity. This can be done through various methods, such as infrared spectroscopy (IR), nuclear magnetic resonance spectroscopy (NMR), and mass spectrometry (MS).
It appears in the form of a white powder.

What it is for and where
Ethyldronic acid is a type of bisphosphonate, a class of drugs that prevent bone density loss. In the context of cosmetics and personal care products, it is used as a chelating agent, helping to prevent the breakage of the product and to maintain its clarity and its color, also preventing the growth of microbes.
In water and detergent treatment, ethyldronic acid helps bind and neutralize metal ions to prevent them from contributing to water hardness or causing discoloration. It is also used in industrial environments to prevent fouling and corrosion.
Dentistry
Prior to obturation, the intracanal medicament should be removed. Medicament residues can reduce root canal permeability by promoting the formation of calcium carbonate particles and interfering with sealing ability of root canal sealers (1).
The aim of this study was to compare the efficiency of irrigation protocols (EDTA, citric acid, and etidronate) in Ca(OH)2 removal. Inter-group comparison showed that in the coronal-third, etidronate (Group-III) showed highest cleanliness scores, followed by EDTA (2).
In this root canal study, ethidronic acid showed less removal efficacy than SmearClear and MTAd (3).
Orthopedics
Hip fracture is among the most common causes of acute immobilization in elderly patients leading to increased bone resorption, and elderly patients with hip fracture are at high risk for a subsequent hip fracture. Etidronate therapy inhibits bone resorption and improves calcium balance, and such therapy may prevent bone loss and reduce the risk of subsequent hip fracture (4).
Cosmetics
It is a restricted ingredient as III/53 a Relevant Item in the Annexes of the European Cosmetics Regulation 1223/2009.
Chelating agent. It has the function of preventing unstable reactions and improving the bioavailability of chemical components within a product, and removes calcium and magnesium cations that can cause cloudiness in clear liquids.
Other uses
Used as a corrosion inhibitor.
Applications and uses:
- Fertilizers
- Pesticides
- Degreasers
- Detergents
- Antibacterial
Synonyms: Etidronate, HEDP, 1-Hydroxyethylidene-1,1-Bisphosphonate
Although in medicine it is used as synonym of Eidronic Acid, Etidronate is a salt of etidronic acid.
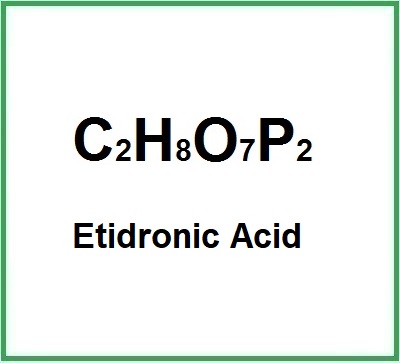
- Molecular formula: C2H8O7P2
- Molecular weight: 206.027
- CAS: 2809-21-4
References__________________________________________________________________
(1) Margelos J, Eliades G, Verdelis C, Palaghias G Interaction of calcium hydroxide with zinc oxide-eugenol type sealers: a potential clinical problem. J Endod. 1997 Jan; 23(1):43-8.
Abstract. When a ZnOE type sealer was placed in root canals treated previously with calcium hydroxide dressing, an accelerated sealer setting rate occasionally occurred. This clinical observation led to the present experimental design aiming to investigate the effect of calcium hydroxide on a ZnOE cement and ZnOE type sealers and to preliminarily assess the removal efficiency of a calcium hydroxide preparation from root canal systems. Micro-MIR FTIR spectroscopy was used to quantify the effect of calcium hydroxide on the setting reactions of a ZnOE cement and two ZnOE type sealers. The removal efficiency of calcium hydroxide from root canal systems was evaluated after treatment with NaOCl; NaOCl and filing; and NaOCl plus EDTA and filing. Calcium hydroxide preferentially interacted with eugenol inhibiting the ZnO-eugenol chelate formation. The Ca(OH)2-eugenol interaction was rapid, and kinetically dependent, leading to residual eugenol in the set product. The set ZnOE cement and the ZnOE type sealers in contact with calcium hydroxide were brittle in consistency and granular in structure. Although none of the treatments tested completely removed calcium hydroxide from root canals, treatment with EDTA significantly reduced the extent of residual calcium hydroxide.
(2) Chockattu SJ, Deepak BS, Goud KM. Comparison of efficiency of ethylenediaminetetraacetic acid, citric acid, and etidronate in the removal of calcium hydroxide intracanal medicament using scanning electron microscopic analysis: An in-vitro study. J Conserv Dent. 2017 Jan-Feb;20(1):6-11. doi: 10.4103/0972-0707.209079.
Abstract. Context: Being integral to root canal therapy, obturation can be performed adequately only after the removal of intracanal medicament. One technique involves the use of chelating agents such as ethylenediaminetetraacetic acid (EDTA) and citric acid. Etidronic acid, a relatively new chelator, has smear layer removal ability and lesser dentinal erosion. It is untested in calcium hydroxide (Ca[OH]2) medicament removal. Aim: The aim of this study was to compare the efficiency of irrigation protocols (EDTA, citric acid, and etidronate) in Ca(OH)2 removal....Conclusion: The solution pH of citric acid and etidronate impacts their Ca(OH)2 removal efficiency in different ways: the highly alkaline pH of Ca(OH)2 increases citric acid pH toward neutrality, where it becomes an inefficient chelator; on the contrary, high acidity of etidronate compensates for its weaker chelation. Etidronate may not require 5 min duration for Ca(OH)2 removal due to the likelihood of dentinal erosion.
(3) Yadav HK, Yadav RK, Chandra A, Tikku AP. A Scanning Electron Microscopic Evaluation of the Effectiveness of Etidronic Acid, SmearClear and MTAD in Removing the Intracanal Smear Layer. J Dent (Shiraz). 2017 Jun;18(2):118-126.
Abstract. Statement of the problem: Root canal therapy should not simply be the extirpation of the pulp and widening of the canal. But one should also focus on how to completely remove the loosely-attached smear layer because it has adverse effects on the final outcome of the treatment.. Purpose: This study compared the efficacy of Etidronic acid, SmearClear and MTAD to remove the smear layer created during instrumentation in different regions of the root canal.....Conclusion: Chelators are essential for complete smear layer removal in association with organic solvent.
(4) Kushida K, Fukunaga M, Kishimoto H, Shiraki M, Itabashi A, Inoue T, Kaneda K, Morii H, Nawata H, Yamamoto K, Ohashi Y, Orimo H. A comparison of incidences of vertebral fracture in Japanese patients with involutional osteoporosis treated with risedronate and etidronate: a randomized, double-masked trial. J Bone Miner Metab. 2004;22(5):469-78. doi: 10.1007/s00774-004-0509-z.
Abstract. To demonstrate the clinical benefit of risedronate at 2.5 mg daily in the treatment of involutional osteoporosis, the effect of risedronate on incidence of vertebral fracture was compared with that of etidronate. A total of 547 patients with one to four vertebral fractures were randomized to receive either treatment with 2.5 mg/day of risedronate or intermittent treatment (treatment of 2 weeks and off period of 10 weeks) with 200 mg/day of etidronate for 96 weeks in a double-masked fashion. All patients received 200 mg calcium supplement daily. Lateral and anteroposterior thoracic and lumbar spine radiographs were obtained at baseline and at 24, 48, 72, and 96 weeks. Cumulative incidence rates of patients who had at least one new or worsening vertebral fracture during the 96-week period were 12.3% for risedronate and 14.2% for etidronate, and it was verified that the fracture prevention effect of risedronate was not inferior to that of etidronate. The incidence rates of fracture during the initial 24-week period were 8.8% for risedronate and 6.0% for etidronate, but the cumulative incidence rate of fracture from 24 to 96 weeks was lower in the risedronate group (3.9%) as compared to the etidronate group (8.7%). Height loss was significantly less in the risedronate group (-0.28 cm) than in the etidronate group (-0.70 cm) after 96 weeks. Decreases in bone resorption markers including urinary total deoxypyridinoline and NTX were significantly greater in the risedronate group than in the etidronate group throughout the treatment period. An improvement of patient QOL was observed in both groups. No significant difference in the incidence of adverse events was observed between the two treatments. Daily oral risedronate (2.5 mg) was shown to provide an effective therapy for involutional osteoporosis in Japanese patients with good tolerability.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips about this object in __Italiano (1)
Component type: Chemical Main substances: Last update: 2019-10-27 19:18:50 | Chemical Risk: |