![]() Citral
Citral
Rating : 7
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
Pros:
Antioxidant (1) Antibacterial (1) Anti-inflammatory (1)10 pts from Ottika11
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.Evaluate | Where is this found? |
| "Descrizione" about Citral Review Consensus 10 by Ottika11 (2059 pt) | 2024-Jul-01 17:58 |
| Read the full Tiiip | (Send your comment) |
Citral (3,7-dimethyl-2,6-octadien-1-al) is a racemic mixture, an acyclic monoterpenoid aldehyde, composed of two geometric terpenoid stereoisomers, geranial and neral.
Citral is a term used to refer to two similar compounds, geranial (Citral A) and Neral (Citral B), which are both components of many essential oils, including citronella and citrus oils. The term "Citral" comes from the Latin word for lemon ("citrus"), which reflects its strong lemon aroma.
The name defines the structure of the molecule:
- Citral is a monoterpenoid, which means that it is derived from two isoprene units. Isoprene is a five-carbon compound, and monoterpenoids are a class of terpenes that consist of two isoprene units, or ten carbon atoms in total.
- The structure of Citral includes a long carbon chain with single and double bonds alternating, characteristic of many terpenes and terpenoids.
- Citral also includes an aldehyde group (-CHO), which contributes to its strong aroma.
- The two forms of Citral, geranial and Neral, are geometric isomers. This means that they have the same molecular formula and sequence of bound atoms, but differ in the orientation of their atoms in space. In the case of geranial and Neral, the difference lies in the position of the double bond adjacent to the aldehyde group.
Citral can be obtained naturally from essential oils such as citronella oil, lemon oil and orange oil. However, it can also be chemically synthesized. Here is a simplified version of the chemical synthesis process:
- Condensation reaction. Citral synthesis begins with a condensation reaction between acetone and acetaldehyde in the presence of a base, such as sodium hydroxide. This reaction forms a compound known as 6-methyl-5-hepten-2-one.
- Oxidation. 6-methyl-5-hepten-2-one is oxidized to form Citral using an oxidizing agent such as chromic acid.
- Purification. The resulting Citral is purified to remove any starting materials and unreacted by-products through distillation, where the Citral is heated and the vapors are collected and condensed.
- Isomerization. Since Citral consists of two isomers, geranial and Neral, the ratio of these two isomers can be regulated through isomerization, which involves heating Citral in the presence of an acid or base.
It occurs as a pale yellow or yellow liquid with an intense lemon aroma and flavour.

Where it is extracted
It is extracted synthetically mainly from citrus fruits, lemongrass (Cymbopogon citratus) and ginger (Zingiber officinale). Other sources are the australian tree Backhousia citriodora F.. Muell.which belongs to the Myrtaceae family and is also called 'lemon myrtle', the oriental plant Litsea cubeba, and the eucalyptus Corymbia citriodora.
What it is used for and where
Medical
In this study, citral demonstrated pro-apoptotic and antiproliferative activity in breast carcinoma cell lines (1), antitumour activity in breast (2) and prostate (3) cancer models.
The scientific literature has long emphasised the importance of citral's anti-inflammatory effects, its inhibitory effects on cytokine production and its role as an immunomodulatory agent (4).
Citral also has potent in vitro antifungal activity against the most common dermatological infections, and the causative organisms include dermatophytes, yeasts and non-dermatophyte filamentous fungi (5).
Some weak diuretic effects have also been shown by citral in oral administrations (6) and, with regard to obesity, it reduced interleukin (IL)-6 levels in the hypothalamus in laboratory animal studies (7).
Cosmetics
Citral is a restricted ingredient as III/70 a Relevant Item in the Annexes of the European Cosmetics Regulation 1223/2009. Ingredient at risk: 3,7-Dimethyl-2,6-octadienal
It is a fragrance, a synthetic perfume that is added to cosmetics, detergents and other products both to perfume the product and to act as an antifungal agent. Raw material for synthesising purple ketone, ionones and methylions.
Fragrance. It plays a decisive and important role in the formulation of cosmetic products as it provides the possibility of enhancing, masking or adding fragrance to the final product, increasing its marketability. It is able to create a perceptible pleasant odour, masking a bad smell. The consumer always expects to find a pleasant or distinctive scent in a cosmetic product.
Perfuming. Unlike fragrance, which can also contain slightly less pleasant or characteristic odours, the term perfume indicates only very pleasant fragrances. Used for perfumes and aromatic raw materials.
Flavoring agent. The purpose of this ingredient is to modify the solution to impart a certain flavour. Natural flavouring extracts are rather expensive, so the cosmetic and pharmaceutical industries resort to synthesised substances that have sensory characteristics mostly similar to natural flavourings or are naturally equivalent. This ingredient is isolated through chemical processes or is synthesised from chemicals. It is also referred to as Aroma.
Typical optimal commercial product characteristics Citral
| Appearance | Pale yellow or yellow-coloured liquid |
| Density | 0.9±0.1 g/cm3 0.888 g/ml |
| Boiling Point | 229.0±9.0°C at 760 mmHg |
| Melting point | < -10ºC |
| Flash point | 101.7±0.0°C |
| Vapor pressure | 0.2 mm Hg ( 200°C) 0.1±0.5 mmHg at 25°C |
| Vapor density | 5 (vs air) |
| Refractive index | n20/D 1.488(lit.) |
| Water | ≤0.5% |
| Acidity(mgKOH/g) | ≤0.5 |
| Sulphated ash | ≤0.5%/g |
| Residue on ignition | ≤0.1% |
| Heavy metals | ≤10 ppm |
| PSA | 17.07000 |
| LogP | 3.17 |
| Safety |  |
 |  |
 |  |
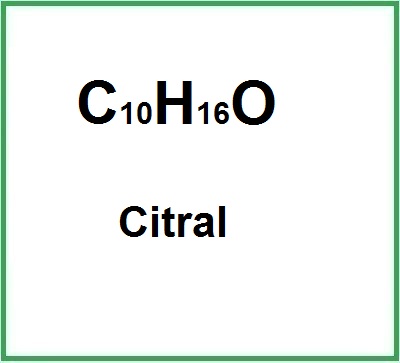
- Molecular Formula C10H16O
- Linear Formula (CH3)2C=CHCH2CH2C(CH3)=CHCHO
- Molecular Weight 152,237 g/mol
- Exact Mass 152.120117
- CAS 5392-40-5 68855-99-2
- UNII 758ZMW724E
- EC Number 226-394-6 290-018-7
- DSSTox Substance ID DTXSID6024836
- IUPAC (2E)-3,7-dimethylocta-2,6-dienal
- InChI=1S/C10H16O/c1-9(2)5-4-6-10(3)7-8-11/h5,7-8H,4,6H2,1-3H3/b10-7+
- InChl Key WTEVQBCEXWBHNA-JXMROGBWSA-N
- SMILES CC(=CCCC(=CC=O)C)C
- MDL number MFCD00006997
- PubChem Substance ID 24893046
- ChEBI 16980
- Beilstein 1721871
- ICSC 1725
- NSC 6170
- RTECS RG5075000
- NACRES NA.22
- UN 2810
- FEMA 2303
- JECFA 1225
- SCHEMBL 23073
Synonyms :
- 2,6-Octadienal, 3,7-dimethyl-
- 3,7-dimethyl-2,6-octadien-1-al
- 2,6-Octadienal,3,7-dimethyl-
- 3,7-Dimethyl-1,2,6-octadienal
- 3,7-Dimethyll-2,6-octadienal
- 3,7-dimethyl-octa-2,6-dienal
- 6-Octadienal,3,7-dimethyl-2
- femanumber2303
- lemsyngb
- NCI-C56348
- Oiloflemongrass
- Neral
- Lemarome
- Lemsyn
- Geranial
- FEMA 2303
- Citrathal
- AKOS NCG1-0113
- Gerznial
References____________________________________________________________________
(1) Chaouki W, Leger DY, Liagre B, Beneytout JL, Hmamouchi M. Citral inhibits cell proliferation and induces apoptosis and cell cycle arrest in MCF-7 cells. Fundam Clin Pharmacol. 2009 Oct;23(5):549-56. doi: 10.1111/j.1472-8206.2009.00738.x.
Abstract. Many natural components of plants extract are studied for their beneficial effects on health and particularly on carcinogenesis chemoprevention. In this study, we investigated the effect of citral (3,7-dimethyl-2,6-octadienal), a key component of essential oils extracted from several herbal plants, on the proliferation rate, cell cycle distribution, and apoptosis of the human breast cancer cell line MCF-7. The effects of this compound were also tested on cyclo-oxygenase activity. Citral treatment caused inhibition of MCF-7 cell growth (IC(50)-48 h: 18 x 10(-5)m), with a cycle arrest in G(2)/M phase and apoptosis induction. Moreover, we observed a decrease in prostaglandin E(2) synthesis 48 h after citral treatment. These findings suggest that citral has a potential chemopreventive effect.
(2) Zeng S, Kapur A, Patankar MS, Xiong MP. Formulation, Characterization, and Antitumor Properties of Trans- and Cis-Citral in the 4T1 Breast Cancer Xenograft Mouse Model. Pharm Res. 2015 Aug;32(8):2548-58. doi: 10.1007/s11095-015-1643-0.
Abstract. Purpose: Citral is composed of a random mixture of two geometric stereoisomers geranial (trans-citral) and neral (cis-citral) yet few studies have directly compared their in vivo antitumor properties. A micelle formulation was therefore developed....Conclusions: Geranial is significantly more potent than neral and citral at 80 mg/kg (p < 0.001) and western blot of tumor tissues confirms that autophagy and not apoptosis is the major mechanism of tumor growth inhibition in p53-null 4T1 cells.
(3) Balusamy SR, Perumalsamy H, Veerappan K, Huq MA, Rajeshkumar S, Lakshmi T, Kim YJ. Citral Induced Apoptosis through Modulation of Key Genes Involved in Fatty Acid Biosynthesis in Human Prostate Cancer Cells: In Silico and In Vitro Study. Biomed Res Int. 2020 Mar 18;2020:6040727. doi: 10.1155/2020/6040727.
(4) Bachiega TF, Sforcin JM. Lemongrass and citral effect on cytokines production by murine macrophages. J Ethnopharmacol. 2011 Sep 1;137(1):909-13. doi: 10.1016/j.jep.2011.07.021.
(5) Silva Cde B, Guterres SS, Weisheimer V, Schapoval EE. Antifungal activity of the lemongrass oil and citral against Candida spp. Braz J Infect Dis. 2008 Feb;12(1):63-6. doi: 10.1590/s1413-86702008000100014.
Abstract. Superficial mycoses of the skin are among the most common dermatological infections, and causative organisms include dermatophytic, yeasts, and non-dermatophytic filamentous fungi. The treatment is limited, for many reasons, and new drugs are necessary. Numerous essential oils have been tested for both in vitro and in vivo antifungal activity and some pose much potential as antifungal agents. By using disk diffusion assay, we evaluated the antifungal activity of lemongrass oil and citral against yeasts of Candida species (Candida albicans, C. glabrata, C. krusei, C. parapsilosis and C. tropicalis). This study showed that lemongrass oil and citral have a potent in vitro activity against Candida spp.
(6) Carbajal D, Casaco A, Arruzazabala L, Gonzalez R, Tolon Z. Pharmacological study of Cymbopogon citratus leaves. J Ethnopharmacol. 1989 Feb;25(1):103-7. doi: 10.1016/0378-8741(89)90049-4.
Abstract. Cymbopogon citratus leaves are employed by the Cuban population as an antihypertensive and anti-inflammatory folk medicine. A 10% or 20% decoction of leaves was tested using arterial pressure in rats, urine production and carrageenan-induced edema in rats. The decoction showed some dose-related hypotensive effects given intravenously and some weak diuretic and anti-inflammatory effect when given orally.
(7) Emílio-Silva MT, Rodrigues VP, Bueno G, Ohara R, Martins MG, Horta-Júnior JAC, Branco LGS, Rocha LRM, Hiruma-Lima CA. Hypothermic Effect of Acute Citral Treatment during LPS-induced Systemic Inflammation in Obese Mice: Reduction of Serum TNF-α and Leptin Levels. Biomolecules. 2020 Oct 17;10(10):1454. doi: 10.3390/biom10101454.
Abstract. Citral is a mixture of monoterpenes present in the essential oil of several plants, such as Cymbopogon citratus and Zingiber officinale, possessing anti-inflammatory, anti-ulcerogenic, and antipyretic actions. We investigated the action of citral on body temperature (Tb) and inflammatory signaling in eutrophic and obese mice during Systemic Inflammation (SI) induced by Lipopolysaccharide (LPS). Thus, we assessed the effect of citral (25, 100, and 300 mg/kg) and ibuprofen in LPS-induced SI in Swiss male mice fed a standard diet (SD) or high-fat diet (HFD) for 12 weeks. Following SI induction, we measured Tb and collected the serum, hypothalamus, and gastric mucosa for biochemical measurements. Acute treatment with citral decreased the Tb of both SD and HFD-fed animals. Citral (300 mg/kg) treatment caused a significantly lower Tb variation in HFD-fed animals than in those fed the SD. Citral reduced peripheral levels of tumor necrosis factor (TNF)-α in SD and HFD mice and decreased serum leptin concentration in HFD mice 90 min after the LPS challenge. Furthermore, citral also reduced interleukin (IL)-6 levels in the hypothalamus of obese mice. In summary, citral effectively reduced Tb during SI by reducing inflammatory mediators with a distinct action profile in HFD mice when compared with SD.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Citral, studies" about Citral Review Consensus 8 by Ottika11 (2059 pt) | 2019-Oct-17 18:59 |
| Read the full Tiiip | (Send your comment) |
Citral is a chemical component found in citrus, fruit and vegetables. In particular it is found in citronella where it is the main component with about 68.78%. It has demonstrated protection against induced oxidative stress and could be a promising agent for lung cancer chemoprevention (1).
Stable and safe for the skin (2), Citral has proved to be an excellent bactericide for use in the treatment of drug-resistant infections (3).
Citral exhibited a strong cytotoxic effect and anti-inflammatory activity in a RAW 264.7 cell line (4).
Citral could be a lead candidate phytomolecule for further developing it into an anti-staphylococcal agent (5).
References__________________
(1) Protective effects of lemongrass essential oil against benzo(a)pyrene induced oxidative stress and DNA damage in human embryonic lung fibroblast cells.
Jiang J, Xu H, Wang H, Zhang Y, Ya P, Yang C, Li F.
Toxicol Mech Methods. 2016 Nov
(2) Effects of Carbopol® 934 proportion on nanoemulsion gel for topical and transdermal drug delivery: a skin permeation study.
Zheng Y, Ouyang WQ, Wei YP, Syed SF, Hao CS, Wang BZ, Shang YH.
Int J Nanomedicine. 2016 Nov
(3) Antimicrobial activity, cytotoxicity and chemical analysis of lemongrass essential oil (Cymbopogon flexuosus) and pure citral.
Adukwu EC, Bowles M, Edwards-Jones V, Bone H.
Appl Microbiol Biotechnol. 2016 Nov
(4) Anti-inflammatory and anti-cancer activity of citral: Optimization of citral-loaded solid lipid nanoparticles (SLN) using experimental factorial design and LUMiSizer®.
Zielińska A, Martins-Gomes C, Ferreira NR, Silva AM, Nowak I, Souto EB.
Int J Pharm. 2018 Dec 20;553(1-2):428-440. doi: 10.1016/j.ijpharm.2018.10.065.
(5) Citral, a monoterpenoid aldehyde interacts synergistically with norfloxacin against methicillin resistant Staphylococcus aureus.
Gupta P, Patel DK, Gupta VK, Pal A, Tandon S, Darokar MP.
Phytomedicine. 2017 Oct 15;34:85-96. doi: 10.1016/j.phymed.2017.08.016.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips about this object in __Italiano (2)
Component type: Chemical Main substances:
Last update: 2024-07-01 17:52:52 | Chemical Risk: |