Malic acid (1) is a dicarboxylic acid found in unripe fruits and produces a pleasant sour taste.
It was discovered in 1785 in an apple by Carl Wilhelm Scheele, a Swedish researcher born in 1742 and passed away in 1786, author of numerous discoveries including manganese, chlorine, tartaric acid, glycerin and lactic acid, which he simply classified it as apple acid.
In 1787, Lavoisier and his group of researchers attributed the name malic acid to this acid.
In 1834 the chemist Théophile Jules Pelouze (1807-1867) distilled it and discovered two compounds: maleic acid and para-maleic acid.
It is also found in the human body, in citric acid.
Industrialmente si presenta in forma di polvere bianca.

What it is used for and where
Food
It is used in the food industry to give foods a sour taste. Ingredient included in the list of European food additives as E296.
Cosmetics
Buffering agent. It is an iingredient that can bring an alkaline or acid solution to a certain pH level and prevent it from changing, in practice a pH stabiliser that can effectively resist instability and pH change.
Cosmetic safety
Malic acid was an irritant in clinical tests, with less irritation observed when the pH of the applied material increased, did not cause reproductive toxicity, proved to be a strong ocular irritant (2)
Medical
Used to treat xerostomia, a disease that reduces saliva emission from salivary glands (3), fibromyalgia, a disorder that causes musculoskeletal pain and fatigue (4)
Supplementation of l-Malic acid within an appropriate range holds significant potential for the prevention and treatment of certain diseases (5).
Malic acid studies
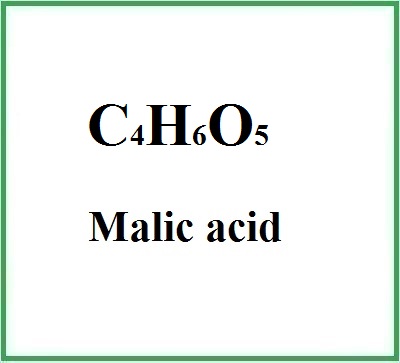
- Molecular Formula: C4H6O5
- Linear Formula HO2CCH2CH(OH)CO2H
- Molecular Weight: 134.087 g/mol
- CAS: 97-67-6 6915-15-7
- EC Number: 202-601-5
- PubChem Substance ID 329750312
- MDL number MFCD00064212
- Beilstein Registry Number 1723539
Synonyms:
- DL-malic acid
- Butanedioic acid, hydroxy-
- 2-Hydroxybutanedioic acid
- 2-Hydroxysuccinic acid
- malate
- Malic acid, DL-
- Musashi-no-Ringosan
- alpha-Hydroxysuccinic acid
- Hydroxybutandisaeure
- dl-Hydroxybutanedioic acid
- Caswell No. 537
- Succinic acid, hydroxy-
- R,S(+-)-Malic acid
- 2-Hydroxyethane-1,2-dicarboxylic acid
- Deoxytetraric acid
- hydroxysuccinic acid
References_________________________________________________________________________
(1) L. Guyton de Morveau, A. Lavoisier, C. Berthollet, A. Fourcroy, Méthode de nomenclature chimique, Cuchet, Paris, 1787, p. 150
(2) Fiume Z. Final report on the safety assessment of Malic Acid and Sodium Malate. Int J Toxicol. 2001;20 Suppl 1:47-55. doi: 10.1080/109158101750300946.
Abstract. Malic Acid functions in cosmetic formulations as a pH adjuster, and Sodium Malate functions as a skin conditioning agent-humectant. Malic Acid is reportedly used in almost 50 cosmetic formulations across a range of product types at low concentrations, whereas Sodium Malate is used in only one. As a pH adjuster, Malic Acid is used at low concentrations. One commercial method of preparing Malic Acid is hydration of fumaric acid or maleic acid, and then purified to limit the amount of the starting material present. Because Malic Acid is a component of the Kreb's cycle, another method is fermentation. Malic Acid was relatively nontoxic in acute toxicity studies using animals. In a chronic oral study, feeding Malic Acid to rats resulted only in weight gain changes and changes in feed consumption. Malic Acid did not cause reproductive toxicity in mice, rats, or rabbits. Malic Acid was a moderate to strong skin irritatant in animal tests, and was a strong ocular irritant. Malic Acid was not mutagenic across a range of genotoxicity tests. Malic Acid was irritating in clinical tests, with less irritation seen as pH of the applied material increased. Patients patch tested with Malic Acid, placed on a diet that avoided foods containing Malic or citric acid, and then challenged with a diet high in Malic and citric acid had both immediate urticarial and delayed contact dermatitis reactions. These data were considered sufficient to determine that Malic Acid and Sodium Malate would be safe at the low concentrations at which these ingredients would be used to adjust pH (even though Sodium Malate is not currently used for that purpose). The data, however, were insufficient to determine the safety of these ingredients when used in cosmetics as other than pH adjusters and specifically, the data are insufficient to determine the safety of Sodium Malate when used as a skin conditioning agent-humectant. The types of data required for the Expert Panel to determine the safety of Sodium Malate as a skin-conditioning agent are: concentration of use data; dermal irritation and sensitization data; and ocular irritation data, if available. The data needed to assess the safety of Malic Acid or Sodium Malate for some function other than as a skin-conditioning agent cannot be specified without knowing the intended function. Were these ingredients to be used as exfoliants, for example, data similar to that included in the Cosmetic Ingredient Review safety assessment of Glycolic Acid would be needed. Until these data are available, it is concluded that the available data are insufficient to support the safety of these ingredients in cosmetic formulations for functions other than use as a pH adjuster.
(3) Liu G, Qiu X, Tan X, Miao R, Tian W, Jing W. Efficacy of a 1% malic acid spray for xerostomia treatment: A systematic review and meta-analysis. Oral Dis. 2023 Apr;29(3):862-872. doi: 10.1111/odi.14116.
(4) Ferreira I, Ortigoza Á, Moore P. Magnesium and malic acid supplement for fibromyalgia. Medwave. 2019 May 28;19(4):e7633. Spanish, English. doi: 10.5867/medwave.2019.04.7632.
Abstract. Introduction: Fibromyalgia is characterized by myalgia and a combination of different symptoms including pain, fatigue, insomnia, morning rigidity, depression and a reduction in every-day functioning. Its aetiology is not clear, but it has been suggested that deficiency in certain minerals such as magnesium may play a role both in the physiopathology and in contributing to the symptoms. Methods: We searched in Epistemonikos, the largest database of systematic reviews in health, which is maintained by screening multiple information sources, including MEDLINE, EMBASE, Cochrane, among others. We extracted data from the systematic reviews, reanalyzed data of primary studies, conducted a meta-analysis and generated a summary of findings table using the GRADE approach. Results and conclusions: We identified seven systematic reviews which included 11 primary studies of which one was a randomized trial. Our conclusion is that the use of magnesium and malic acid in patients with fibromyalgia makes little or no difference on pain and on depressive symptoms.
(5) Lu J, Zhang S, Wu S, Gao C. l-malic acid: A multifunctional metabolite at the crossroads of redox signaling, microbial symbiosis, and therapeutic innovation. Arch Biochem Biophys. 2025 Oct;772:110554. doi: 10.1016/j.abb.2025.
![]() Malic acid
Malic acid