App Tiiips monitors calories, dangerous ingredients
Free! Download now from Play Store!


App Tiiips monitors calories, dangerous ingredients
Free! Download now from Play Store!


| "Club Salute Fluidoflu REVIEW" about Club Salute Fluidoflu Review Consensus 17 by AColumn (9300 pt) | 2020-Apr-14 18:42 |
| Read the full Tiiip | (Send your comment) |
Fluidoflu is a dietary supplement in 14 sachets that contain:
Maltodextrin, sorbitol, N-acetylcysteine, aroma, Vitamin C, bromelain, citric acid, ederagenin, MicroActive® resveratrol, zinc citrate, sucralose, silicon dioxide.
Club Salute is a company that distributes dietary supplements created by a group of pharmacists based in Milan.
Fluidoflu is sold as a dietary supplement with sweetener, in sachets for "Seasonal Winter Remedies".
We examine the ingredients in descending order of quantity on the label and check what we will take with this supplement:
Maltodextrin is a safe component used in countless applications: viscosity agent, preservative, improves product consistency, counteracts the action of Acinetobacter baumannii and Staphylococcus aureus, important nosocomial pathogens. it is also used as a low-calorie softener.
Sorbitol is an artificial sweetener with a sweetening power of about 60% of sugar and a caloric power of about 40% less than sugar. It is obtained industrially by a chemical process from glucose. It also has other interesting qualities such as anti-mould, anti-yeast and preservative.
N-acetylcysteine (it is present with 300mg in 1 sachet) is a compound of acetylated cysteine used for a long time as an adjuvant in bronchitis in the role of fluidifying agent and expectorant of excessive mucus as well as to combat the toxicity of some components such as doxorubicin, paracetamol, acute respiratory distress syndrome. It is included in many formulas of food supplements for its established role as an antioxidant and anti-inflammatory.
Aroma is a flavouring chemical component.
Bromelain (it is present with 100mg in 1 sachet) is a biomolecule, an enzyme that has many properties: anti-inflammatory, antiarthritic, antitumor.
Vitamin C or ascorbic acid, (it is present with 80mg in 1 sachet) is an antioxidant. The body is not able to produce vitamin C on its own and does not store it. It is therefore important to take a certain amount of vitamin C with foods that contain it, daily. It is used in the treatment of colds and flu.
Citric acid, an acid obtained from lemon juice and other fruits, chard juice and other vegetables. It is used as an excipient in pharmaceutical products as an anticoagulant and preservative.
Resveratrol (it is present with 10mg in 1 sachet) is a natural component, a polyphenolic compound found in nature mainly in the skin and seeds of grapes and peanuts. It is an antioxidant.
Ederagenin (present with 0.6mg in 1 sachet) is a saponin and the scientific literature attributes it a wide range of properties such as anti-inflammatory, antidepressant, hepatoprotective.
Zinc citrate is a chemical compound and is used as a dietary supplement in cases of zinc deficiency. Zinc in the human body is an important immunological barrier and its deficiency can lead to skin, bone and eye diseases. It also has antioxidant properties.
Sucralose is obtained industrially by a chemical process from glucose. It's an artificial sweetener.
Silicon dioxide or colloidal silica is an anti-caking agent, prevents lumps from forming in the dough. It must not exceed 2% of the total weight of the product in which it is inserted.
Comment
The ingredients are carefully chosen and targeted to the most common winter sicknesses such as colds, coughs. In particular N-acetylcysteine plays the primary role as a fluidifying agent.I am surprised to see in third place, in order of quantity, Aroma, pure chemistry that I hope can be replaced with a natural flavouring. Sucralose also seems to me unnecessary because Sorbitol performs very well the function of sweetener.
Conclusion
Great product, but I'd prefer without Aroma.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "neoBorocillina Gola Dolore REVIEW" about neoBorocillina Gola Dolore 16 pastiglie Review Consensus 8 by AColumn (9300 pt) | 2020-Apr-04 17:04 |
| Read the full Tiiip | (Send your comment) |
neoBorocillina Gola Dolore is an over-the-counter medicine that can be purchased without a prescription and is indicated for the treatment of symptoms of sore throat.
The package contains 16 tablets.
It's a medication that would be good to take at first symptoms. It is usually effective on the first day of use.
Like all medicines, it can give side effects that are detailed in the package leaflet.
Now let's examine the ingredients that we will put into the body, sorted by decreasing amounts, as indicated on the label:
Flurbiprofene Flurbiprofen is a water-insoluble anti-inflammatory and non-steroidal analgesic drug, is used at low dosages and is commonly found in over-the-counter products both in tablets and sprays. It is a fluorinated drug with potential applications in a large number of diseases and the most common formulation is 8.75mg for use as a symptomatic relief of pharyngitis of viral, bacterial or unknown etiology.
Glicerolo dibeenato Glycerol dibeenate is glycerol incorporated into the formula as a stabilizing additive.
Acesulfame K is the additive E950 used in the food industry as a sugar substitute sweetener and belongs to the non-nutritive sweeteners. It has an Acceptable Daily Intake or ADI of 15 mg/kg body weight.
Aroma limome miele. Instead of including natural active ingredients of lemon and honey, the manufacturer preferred to add chemical components that imitate the taste of lemon and honey.
Isomalto Isomalt is an oligosaccharide or simple sugar.
Copovidone is a synthetic polymer used in the pharmaceutical industry to disperse and suspend the release of the drug.
Conclusion
Good product that has an effect on sore throat. I do not like the inclusion of flavors.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Flurbiprofen studies" about Flurbiprofen Review Consensus 8 by AColumn (9300 pt) | 2022-Jul-31 17:41 |
| Read the full Tiiip | (Send your comment) |
Updates on Flurbiprofen concern use, interactions with other drugs, contraindications.
Khizer Z, Nirwan JS, Conway BR, Ghori MU. Okra (Hibiscus esculentus) gum based hydrophilic matrices for controlled drug delivery applications: Estimation of percolation threshold [published online ahead of print, 2020 Mar 30]. Int J Biol Macromol. 2020;S0141-8130(20)32814-2. doi:10.1016/j.ijbiomac.2020.03.227
Abstract. This study aims to explore the potential of gum extracted from okra fruit (Hibiscus esculentus) in developing hydrophilic matrices for controlled drug release applications, including determination of its percolation threshold. Flurbiprofen (poorly soluble), theophylline (sparingly soluble) and metformin (freely soluble) were employed as model drugs and incorporated using direct compression into matrices containing 40% w/w of three drugs with different physicochemical properties.
Yang Z, Song Q, Cao Z, et al. Design, synthesis and evaluation of flurbiprofen-clioquinol hybrids as multitarget-directed ligands against Alzheimer's disease. Bioorg Med Chem. 2020;28(7):115374. doi:10.1016/j.bmc.2020.115374
Abstract. A series of novel flurbiprofen-clioquinol hybrids were designed and synthesized as multifunctional agents for Alzheimer's disease therapy, and their potential was evaluated through various biological experiments.
Wang D, Yang SK, Zhao MX, et al. Low dose of flurbiprofen axetil decrease the rate of acute kidney injury after operation: a retrospective clinical data analysis of 9915 cases. BMC Nephrol. 2020;21(1):52. Published 2020 Feb 14. doi:10.1186/s12882-020-1711-5
Abstract. Flurbiprofen axetil (FA) is a commonly prescribed agent to relieve perioperative pain, but the relationship between FA and postoperative acute kidney injury (AKI) remains unclear. This study attempted to evaluate the effects of different dose of perioperative FA on postoperative AKI.
Caban-Toktas S, Sahin A, Lule S, et al. Combination of Paclitaxel and R-flurbiprofen loaded PLGA nanoparticles suppresses glioblastoma growth on systemic administration. Int J Pharm. 2020;578:119076. doi:10.1016/j.ijpharm.2020.119076
Abstract. Malignant gliomas are highly lethal. Delivering chemotherapeutic drugs to the brain in sufficient concentration is the major limitation in their treatment due to the blood-brain barrier (BBB). Drug delivery systems may overcome this limitation and can improve the transportation through the BBB. Paclitaxel is an antimicrotubule agent with effective anticancer activity but limited BBB permeability. R-Flurbiprofen is a nonsteroidal antienflammatory drug and has potential anticancer activity. Accordingly, we designed an approach combining R-flurbiprofen and paclitaxel and positively-charged chitosan-modified poly-lactide-co-glycolic acid (PLGA) nanoparticles (NPs) and to transport them to glioma tissue.
Alalwani A, Buhara O, Tüzüm MŞ. Oral Health-Related Quality of Life and the Use of Oral and Topical Nonsteroidal Anti-Inflammatory Drugs for Pericoronitis. Med Sci Monit. 2019;25:9200–9206. Published 2019 Dec 3. doi:10.12659/MSM.918085
Abstract. BACKGROUND. Pericoronitis is inflammation of the tissue surrounding a third molar, or wisdom tooth. This study aimed to evaluate the effects of oral and topical analgesic nonsteroidal anti-inflammatory drugs (NSAIDs) on oral health-related quality of life (OHQoL), in terms of oral health and lifestyle, in patients with symptomatic pericoronitis. MATERIAL AND METHODS. The study included 60 patients who presented with pericoronitis and who did not undergo surgery within the following seven days...
Richy F, Rabenda V, Mawet A, Reginster JY. Flurbiprofen in the symptomatic management of rheumatoid arthritis: a valuable alternative. Int J Clin Pract. 2007 Aug;61(8):1396-406. doi: 10.1111/j.1742-1241.2007.01452.x.
Abstract. The withdrawal of certain cyclooxygenase-2 selective drugs and the availability of over-the-counter non-steroidal anti-inflammatory drugs (NSAIDs) have increased the pressure for researching and prescribing conventional NSAIDs with a favourable efficacy/tolerance ratio in inflammatory diseases, particularly rheumatoid arthritis. The aim of this comprehensive meta-analysis was to evaluate the absolute and relative efficacy and safety of flurbiprofen in the management of rheumatoid arthritis...
Yilmaz Ö, Bayer B, Bekçi H, et al. Synthesis, Anticancer Activity on Prostate Cancer Cell Lines and Molecular Modeling Studies of Flurbiprofen-Thioether Derivatives as Potential Target of MetAP (type II) [published online ahead of print, 2019 Jun 13]. Med Chem.
Abstract. Prostate cancer is still one of the serious causes of mortality and morbidity in men. Despite recent advances in anticancer therapy, there is a still need of novel agents with more efficacy and specificity in the treatment of prostate cancer. Because of its function on angiogenesis and overexpression in the prostate cancer, methionine aminopeptidase-2 (MetAP-2) has been a potential target for novel drug design recently.
Contraindications
Zhao X, Ji L. Flurbiprofen axetil: Analgesic effect and adverse reaction. Pak J Pharm Sci. 2018;31(3(Special)):1163–1167.
Abstract. Flurbiprofen axetil is a targeted analgesic and non steroidal analgesic with lipid microspheres as drug carrier. It can selectively accumulate in surgical incision and reduce the allodynia a caused by surgical trauma. In this paper, the experimental subjects were divided into three groups to analyze the difference in the analgesic effect of different doses of flurbiprofen axetil for postoperative analgesia. The patients in group A, B and C were treated with flurbiprofen axetil injection 100, 150, 200mg, respectively. The results showed that MAP, HR, static and dynamic VAS scores and postoperative pain incidence in group B and group C were lower than those in group A, and B group had better analgesic effect and lower incidence of adverse reactions. In the future, we should continue to explore the correlation between the effect of flurbiprofen axetil for postoperative analgesia on coagulation function and the range of dose safety.
Yi EM, Leech E. Topical flurbiprofen toxicosis in a cat. J Vet Emerg Crit Care (San Antonio). 2017;27(6):707–712. doi:10.1111/vec.12638
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Flurbiprofen Review Consensus 8 by AColumn (9300 pt) | 2023-Sep-11 10:23 |
| Read the full Tiiip | (Send your comment) |
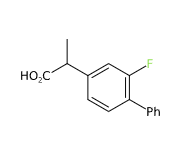
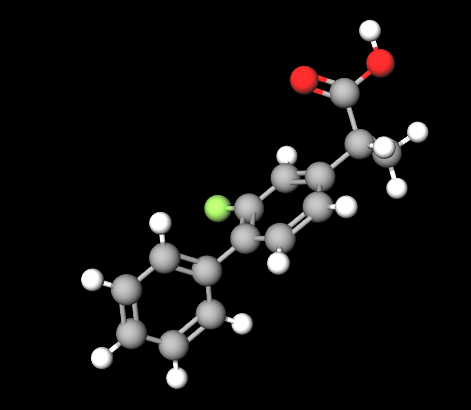
Flurbiprofen is a derivative of propionic acid and.
It appears as white powder insoluble in water.

What it is used for and where
Medical
Take only under medical supervision
It is a non-steroidal anti-inflammatory analgesic drug (NSAID) water insoluble used as a moderate pain reliever, antipyretic and to treat symptoms of chronic arthritis.
It's a fluorinated drug with potential applications in a large number of pathologies and it is used at low dosage and is commonly found in over-the-counter products both in tablets and sprays. The most common formulation is 8.75mg for use as a symptomatic relief of viral, bacterial or unknown aetiology pharyngitis.
In research on patients undergoing intravenous sedation in oral and maxillo-facial surgery, flurbiprofen axetil was found to be the second most common effective and safe sedation method (1).
by suppressing the inflammatory response of the host, and not only its mediators such as prostanoids, cytokines, Flurbiprofen has the ability to alter the progression of periodontitis but also inhibits osteoclast activity and bone resorption (2).
Flurbiprofen axetil might reduce postoperative delirium in patients over 70 years undergoing major noncardiac surgery (3).
Like all drugs it can cause side effects. Always ask the physician.
| Appearance | White powder |
| Boiling Point | 376.2±30.0°C at 760 mmHg |
| Melting Point | 110-112°C(lit.) |
| Flash Point | 181.3±24.6°C |
| Density | 1.2±0.1 g/cm3 |
| PSA | 37.30000 |
| LogP | 4.11 |
| Refraction Index | 1.568 |
| Vapor Pressure | 0.0±0.9 mmHg at 25°C |
| Safety |  |
 |  |
 |  |
Synonyms:
References____________________________________________________
(1) Wang F, YY, Guan M, et al. Pechino Da Xue Xo Yi Xue Ban. 2020;52(1):181–186.
(2) Williams RC, Jeffcoat MK, Howell TH, Rolla A, Stubbs D, Teoh KW, Reddy MS, Goldhaber P. Altering the progression of human alveolar bone loss with the non-steroidal anti-inflammatory drug flurbiprofen. J Periodontol. 1989 Sep;60(9):485-90.
(3) Wang X, Wang Y, Hu Y, et al. Effect of flurbiprofen axetil on postoperative delirium for elderly patients. Brain Behav. 2019;9(6):e01290. doi:10.1002/brb3.1290
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Diphenhydramine hydrochloride insig" about Diphenhydramine hydrochloride Review Consensus 8 by AColumn (9300 pt) | 2023-Feb-14 21:37 |
| Read the full Tiiip | (Send your comment) |
Diphenhydramine hydrochloride insights relate to fields of application and possible contraindications.
Okuno T, Morimoto S, Nishikawa H, et al. Bitterness-Suppressing Effect of Umami Dipeptides and Their Constituent Amino Acids on Diphenhydramine: Evaluation by Gustatory Sensation and Taste Sensor Testing. Chem Pharm Bull (Tokyo). 2020;68(3):234–243. doi:10.1248/cpb.c19-00736
Abstract. Diphenhydramine, a sedating antihistamine, is an agonist of human bitter taste receptor 14 (hTAS2R14). Diphenhydramine hydrochloride (DPH) was used as a model bitter medicine to evaluate whether the umami dipeptides (Glu-Glu and Asp-Asp) and their constituent amino acids (Glu, Asp) could suppress its bitterness intensity, as measured by human gustatory sensation testing and using the artificial taste sensor. Various concentrated (0.001-5.0 mM) Glu-Glu, Asp-Asp, Glu and Asp significantly suppressed the taste sensor output of 0.5 mM DPH solution in a dose-dependent manner. The effect of umami dipeptides and their constituent amino acids was tending to be ranked as follows, Asp-Asp > Glu-Glu >> Gly-Gly, and Asp > Glu >> Gly (control) respectively. Whereas human bitterness intensity of 0.5 mM DPH solution with various concentrated (0.5, 1.0, 1.5 mM) Glu-Glu, Asp-Asp, Glu and Asp all significantly reduced bitterness intensity of 0.5 mM DPH solution even though no statistical difference was observed among four substances. The taste sensor outputs and the human gustatory sensation test results showed a significant correlation. A surface plasmon resonance study using hTAS2R14 protein and these substances suggested that the affinity of Glu-Glu, Asp-Asp, Glu and Asp for hTAS2R14 protein was greater than that of Gly-Gly or Gly. The results of docking-simulation studies involving DPH, Glu-Glu and Asp-Asp with hTAS2R14, suggested that DPH is able to bind to a space near the binding position of Glu-Glu and Asp-Asp. In conclusion, the umami dipeptides Glu-Glu and Asp-Asp, and their constituent amino acids, can all efficiently suppress the bitterness of DPH.
Dahal A, Neupane R, Boddu SH, Renukuntla J, Khupse R, Dudley R. Percutaneous Absorption of Lorazepam, Diphenhydramine Hydrochloride, and Haloperidol from ABH Gel. Int J Pharm Compd. 2020 Mar-Apr;24(2):168-175.
Abstract. The objective of this project was to study the percutaneous absorption of lorazepam, diphenhydramine hydrochloride, and haloperidol from a topical Pluronic lecithin organogel, also known as ABH gel, across the porcine ear skin and verify its suitability for topical application. ABH gel was prepared using lecithin in isopropyl palmitate solution (1:1) as an oil phase and 20% w/v Poloxamer 407 solution as an aqueous phase. The gel was characterized for pH, viscosity, drug content, and thermal behavior. A robust high-performance liquid chromatography method was developed and validated for simultaneous analysis of lorazepam, diphenhydramine hydrochloride, and haloperidol. The percutaneous absorption of lorazepam, diphenhydramine hydrochloride, and haloperidol from ABH gel was carried out using Franz cells across the Strat-M membrane and pig ear skin. The pH of ABH gel was found to be 5.66 ± 0.13. The retention time of diphenhydramine hydrochloride, haloperidol, and lorazepam was found to be 5.2 minutes, 7.8 minutes, and 18.9 minutes, respectively. The ABH gel was found to be stable for up to 30 days. Theoretical steady state plasma concentrations (CSS) of diphenhydramine hydrochloride, haloperidol, and lorazepam calculated from flux values were found to be 1.6 ng/mL, 0.13 ng/mL, and 2.30 ng/mL, respectively. The theoretical CSS of diphenhydramine hydrochloride, haloperidol, and lorazepam were much lower than required therapeutic concentrations for antiemetic activity to relieve chemotherapy-induced nausea and vomiting. From the percutaneous absorption data, it was evident that ABH gel failed to achieve required systemic levels of lorazepam, diphenhydramine hydrochloride, and haloperidol following topical application. Copyright© by International Journal of Pharmaceutical Compounding, Inc
Melnikova M, Wauer US, Mendus D, et al. Diphenhydramine increases the therapeutic window for platinum drugs by simultaneously sensitizing tumor cells and protecting normal cells . Mol Oncol. 2020;10.1002/1878-0261.12648. doi:10.1002/1878-0261.12648
Abstract. Platinum-based compounds remain a well-established chemotherapy for cancer treatment despite their adverse effects which substantially restrict the therapeutic windows of the drugs. Both the cell type-specific toxicity and the clinical responsiveness of tumors have been associated with mechanisms that alter drug entry and export. We sought to identify pharmacological agents that promote cisplatin (CP) efficacy by augmenting the levels of drug-induced DNA lesions in malignant cells and simultaneously protecting normal tissues from accumulating such damage and from functional loss. Formation and persistence of platination products in the DNA of individual nuclei were measured in drug-exposed cell lines, in primary human tumor cells and in tissue sections using an immunocytochemical method. Using a mouse model of CP-induced toxicity, the antihistaminic drug diphenhydramine (DIPH) and two methylated derivatives decreased DNA platination in normal tissues and also ameliorated nephrotoxicity, ototoxicity, and neurotoxicity. In addition, DIPH sensitized multiple cancer cell types, particularly ovarian cancer cells, to CP by increasing intracellular uptake, DNA platination, and/or apoptosis in cell lines and in patient-derived primary tumor cells. Mechanistically, DIPH diminished transport capacity of CP efflux pumps MRP2, MRP3, and MRP5 particularly in its C2+C6 bimethylated form. Overall, we demonstrate that DIPH reduces side effects of platinum-based chemotherapy and simultaneously inhibits key mechanisms of platinum resistance. We propose that measuring DNA platination after ex vivo exposure may predict the responsiveness of individual tumors to DIPH-like modulators. © 2020 The Authors. Published by FEBS Press and John Wiley & Sons Ltd.
Kuruvilla M, Sexton M, Wiley Z, Langfitt T, Lynde GC, Wolf F. A Streamlined Approach to Optimize Perioperative Antibiotic Prophylaxis in the Setting of Penicillin Allergy Labels . J Allergy Clin Immunol Pract. 2019;S2213-2198(19)31050-5.
Abstract. Background: Patients with penicillin allergy labels often receive alternative antibiotics for perioperative prophylaxis, as opposed to first-line cephalosporins (cefazolin/cefuroxime). Provider misconceptions about the risk of cross-reactivity likely drive this prescribing behavior, which is problematic because of its association with increased risk of surgical-site infections. Objective: To develop, implement, and assess the safety of a streamlined approach to perioperative antibiotic selection for surgical patients with a penicillin allergy label, to reduce the use of second-line antibiotics....Conclusions: Using a streamlined algorithm, we were able to significantly reduce the use of second-line antibiotics in penicillin-allergic surgical patients without severe adverse reactions. Copyright © 2019 American Academy of Allergy, Asthma & Immunology. Published by Elsevier Inc.
Li YY, Zeng YS, Chen JY, et al. Prophylactic diphenhydramine attenuates postoperative catheter-related bladder discomfort in patients undergoing gynecologic laparoscopic surgery: a randomized double-blind clinical study. J Anesth. 2020;34(2):232–237. doi:10.1007/s00540-019-02724-3
Abstract. Background: To evaluate the effectiveness of diphenhydramine, an antihistamine with anti-muscarinic properties, for prevention of postoperative catheter-related bladder discomfort (CRBD)....Conclusion: Prophylactic diphenhydramine 30 mg at induction of general anesthesia reduced the incidence and severity of postoperative bladder discomfort without significant side effects in patients receiving gynecologic laparoscopic surgery.
Fein MN, Fischer DA, O'Keefe AW, Sussman GL. CSACI position statement: Newer generation H1-antihistamines are safer than first-generation H1-antihistamines and should be the first-line antihistamines for the treatment of allergic rhinitis and urticaria. Allergy Asthma Clin Immunol. 2019;15:61. Published 2019 Oct 1. doi:10.1186/s13223-019-0375-9
Abstract. Oral H1-antihistamines (AHs) are the most commonly used therapy to treat allergic rhinitis and chronic urticaria. Older, first-generation AHs (e.g. diphenhydramine, hydroxyzine) have significant and common side effects including sedation, impairment with decreased cognitive function, poor sleep quality, dry mouth, dizziness, and orthostatic hypotension. These drugs have also been found to result in death from accidents, intentional or unintentional overdoses, and sudden cardiac death. The unfavourable risk-benefit profile of first-generation AHs led to the development of newer, less-sedating second- and third-generation AHs, which first became available in Canada in the 1980s. High-quality trials have proven that newer generation AHs are superior in safety compared to older first-generation AHs. On average, they have improved potency and efficacy. Second- and third-generation AHs are the recommended first-line treatment for mild allergic rhinitis and acute and chronic urticaria. Despite this evidence, older first-generation AHs continue to be over-utilized because of their over-the-counter (OTC) status and long history of use. The Canadian Society of Allergy Clinical Immunology (CSACI) recommends that newer generation AHs should be preferred over first-generation AHs for the treatment of allergic rhino-conjunctivitis and urticaria. To promote this recommendation, education of health professionals and the public is necessary. Further, given the dangers of older first-generation AHs, we believe they should be used only as a last resort with eventual consideration given to having them only available behind the counter in pharmacies. © The Author(s) 2019.
Sabins D, Diep T, McCartan P, Patel S, Zhao F. Stability and Compatibility of Diphenhydramine Hydrochloride in Intravenous Admixtures: A New Look at an Old Drug. Hosp Pharm. 2019;54(5):330–334. doi:10.1177/0018578718802586
Abstract. Purpose: Intravenous (IV) admixtures of diphenhydramine are widely used in hospitalized patients to prevent or treat hypersensitivity reactions. However, there is limited data to support the admixture preparation in this manner. This study was designed to investigate the stability and compatibility of diphenhydramine in IV admixtures with a goal to establish a 14-day beyond-use dating with storage under refrigeration. Methods: The commercially available 50 mg/mL diphenhydramine hydrochloride injection vials were used to prepare the 0.2 and 1.0 mg/mL IV admixtures in 0.9% sodium chloride injection and 5% dextrose injection in 50 mL polyvinyl chloride (PVC) bags. The IV bags were sealed and stored under refrigeration (2°C-8°C) for the stability study. At each predetermined time point, samples were taken for visual inspection, pH measurement, and analysis by a stability-indicating high-performance liquid chromatography (HPLC) method. Results: The freshly prepared IV admixtures appeared clear, colorless, and particulate-free with pH readings of 4.44 to 4.60. The initial drug concentrations of all samples were confirmed by HPLC to be within 101.8% to 103.6% of the label claims. Over the 14 days of the study period, there was no significant change in the appearance or pH values for all stability samples. The HPLC results also confirmed that there was no more than ±2% change of the initial drug concentration in any stability samples. Conclusion: Diphenhydramine hydrochloride IV admixtures of 0.2 and 1.0 mg/mL are compatible with 0.9% sodium chloride injection and 5% dextrose injection in PVC bags. These IV admixtures are stable chemically and physically for up to 14 days when stored under refrigeration (2°C-8°C).
Pan W, Cao Z, Liu D, Jiao Y. Protective Effect of Diphenhydramine against Traumatic Brain Injury in Rats via Modulation of Oxidative Stress and Inflammation. Pharmacology. 2020;105(1-2):47–53. doi:10.1159/000502767
Abstract. Background: Traumatic brain injury (TBI) is considered a major burden across the globe affecting both individuals and their families. Therefore, the present study was conducted to determine the protective effect of diphenhydramine (DPM) against TBI in experimental rats....Results: Results of the study suggest that DPM causes reduction in CE and prevents neuronal degeneration. It also causes reduction in inflammation and oxidative stress in a dose-dependent manner. The level of Bax was found to be elevated, together with reduction in the Bcl-2 level in the DPM-treated group. Conclusion: DPM exerts a neuroprotective effect after TBI via the attenuation of oxidative stress, inflammation, and mitochondrial apoptosis pathways. © 2019 S. Karger AG, Basel.
Nguyen T, Polyakova B, Cerenzio J, Ramilo JR. Diphenhydramine Use in End-Stage Kidney Disease . Am J Ther. 2019;10.1097/MJT.0000000000001057. doi:10.1097/MJT.0000000000001057
Abstract. Background: Diphenhydramine is commonly used in patients with kidney disease and end-stage kidney disease (ESKD) for sleep, allergic reactions, itching, and dialysis treatment related complications, and misuse associated with diphenhydramine is also reported. Diphenhydramine's pharmacokinetics property is reviewed and discussed....Results: There is lack of studies available for diphenhydramine, kidney disease, and dialysis. There were case reports of diphenhydramine abuse and toxicity due to overdose. Diphenhydramine is highly bound to protein that limits its ability to dialyze, and therefore, it may predispose to side effects. Information on diphenhydramine used in the dialysis population is scarce, and dosing toxicity is unknown. Conclusions: The data available for use of diphenhydramine in ESKD and dialysis are limited. Clinicians should use caution with the use of diphenhydramine in this population. Copyright © 2019 Wolters Kluwer Health, Inc.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Diphenhydramine hydrochloride Review Consensus 8 by AColumn (9300 pt) | 2023-Sep-11 10:59 |
| Read the full Tiiip | (Send your comment) |
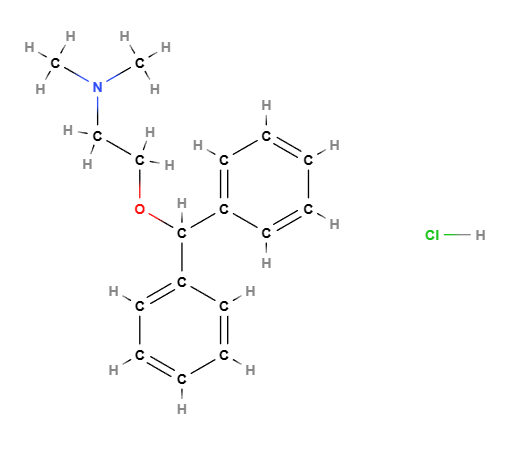
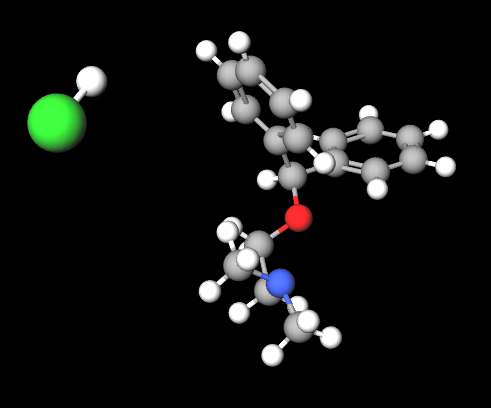
Diphenhydramine hydrochloride is a long-standing drug as it was marketed in 1946 and has been used primarily for the treatment of allergies.
What it is for and where
Medical
Take only under medical supervision
It is a sedative antihistamine, a known agonist of 14 (hTAS2R14) and functional antagonist of the D2 receptor, it is an antidopaminergic agent that has the function of blocking post-synaptic dopamine receptors by exerting a control on symptoms such as headache (1), nausea (2), transient insomnia (3).
In particular, insomnia is a disorder that affects about 30% of the world's population and consists of difficulty falling asleep, difficulty maintaining sleep, and a feeling of not having slept well. These disorders can lead to more complex pathologies such as depression, anxiety, intolerance for life. Those who suffer from insomnia can be affected by very dangerous daytime sleepiness when driving motor vehicles. Diphenhydramine hydrochloride and melatonin have proved to be the most popular over-the-counter drugs. Scientific opinions are conflicting in this respect (4) (5).
Diphenhydramine hydrochloride administered intravenously in endoscopic procedures such as colonoscopy is useful in patients who are difficult to sedate (6).
Like all drugs it can cause side effects. Always ask the physician.
 |  |
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Actigrip giorno & notte Review Consensus 9 by AColumn (9300 pt) | 2023-Feb-10 21:05 |
| Read the full Tiiip | (Send your comment) |
It is a drug that you should take at first symptoms. It is usually effective after the first day of use.
Like all medicines, it can give side effects which are detailed on pages 4 and 5 of the package leaflet.
Now let's examine the ingredients that we put into the body, sorted by decreasing amounts, as indicated on the label:
Well, now we know what we ingested, so my doctor's advice is to use this medicine only in case of actual need and not to exceed the recommended doses at all.
Personally, it stopped my cold and nasal congestion in a couple of days, but I felt the effects immediately from day one. However, it left me with a cough that I had to treat with other medicines. In my body, for decades, I have always had these symptoms: first a slight sore throat, then a cold and then a cough. I rarely had a fever. In any case Actigrip day & night, as we have seen, is both an antipyretic (takes away fever) and a nesal decongestant.
Excellent product, but use only when strictly necessary.
The price is very different: if you buy it on the internet it is between 8 and 9 euros. In pharmacies you can find it between 13 and 14.35 euros.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips in __Italiano (65)
