| "Descrizione" by Frank123 (11988 pt) | 2026-Feb-12 12:26 |
Review Consensus: 10 Rating: 10 Number of users: 1
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
E1414: proprietà, usi, pro, contro, sicurezza
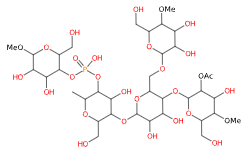
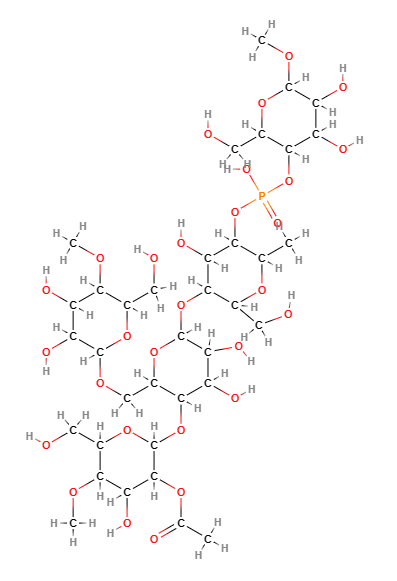
E1414 (Fosfato di diamido acetilato) è un amido modificato ottenuto trattando amidi alimentari con acetilazione e fosfatazione per migliorarne le prestazioni tecnologiche.
In pratica serve a rendere l’amido più stabile durante lavorazioni aggressive, come riscaldamento prolungato, shear, surgelazione e scongelazione. Agisce come addensante e stabilizzante, aumentando la viscosità e contribuendo a una texture più uniforme e ripetibile tra lotti.
Riduce la tendenza a sineresi e separazione di fase, migliorando la tenuta di salse, creme, ripieni e prodotti a base di latte o vegetali. La modifica chimica controlla anche la retrogradazione dell’amido, aiutando a mantenere consistenza e “morbidezza” nel tempo.
È utile in prodotti con cicli termici o shelf-life lunga perché offre maggiore tolleranza a variazioni di processo e conservazione. Dal punto di vista applicativo si integra bene in matrici acquose e miste, con resa più prevedibile rispetto ad amidi nativi.
In etichetta compare come additivo E1414 e il suo impiego è regolato da criteri e limiti che dipendono dalla categoria alimentare e dalla normativa vigente.
Si presenta in forma di polvere bianca o granuli bianchi.

A cosa serve e dove si usa
Alimentazione
Ingrediente inserito nella lista degli additivi alimentari europei come E1414 con funzione di stabilizzante, addensante per fornire consistenza e conservazione anche in cibi ad alta acidità. E' utilizzato per ottenere una qualità migliore in prodotti alimentari sterilizzati a basso valore di pH. Utile nella produzione di maltodestrine, è tuttavia piuttosto suscettibile alla degradazione enzimatica (1).
Sicurezza
Il Gruppo di esperti scientifici dell'EFSA sugli additivi alimentari e le fonti di nutrienti aggiunti agli alimenti ritiene che non vi sia alcun problema di sicurezza per l'uso di amidi modificati come additivi alimentari agli usi e ai livelli d'uso dichiarati per la popolazione generale e che non è necessaria una DGA numerica (2).
 |  |
 |  |
- Formula molecolare C36H63O29P
- Peso molecolare 990.8
- CAS 68130-14-3
- UNII
- EC Number 813-662-8
Bibliografia_____________________________________________________________________
(1) Pycia, K., Juszczak, L., Gałkowska, D., Socha, R. and Jaworska, G., 2017. Maltodextrins from chemically modified starches. Production and characteristics. Starch‐Stärke, 69(5-6), p.1600199.
Abstract. The aim of this work was to evaluate whether the type of chemically modified starch (cross-linked and/or stabilised) has any effect on its susceptibility to α-amylase and on the physicochemical properties of the resulting maltodextrins of dextrose equivalent (DE) of 6, 11 and 16. The duration of the enzymatic hydrolysis of starch was established experimentally on the basis of a course of individual starch hydrolysis. The type of chemically modified starch was found to have a significant effect on its susceptibility to enzymatic hydrolysis. Native potato starch was more easily hydrolysed than acetylated starch and acetylated distarch adipate. The types of starch most susceptible to enzymatic degradation were distarch phosphate and acetylated distarch phosphate. The results of a chromatographic analysis revealed that the maltodextrins of DE 6 differed from one another significantly in terms of the amounts of each of the DP1–DP5 fractions. The number average molecular weight and the weight average molecular weight of the maltodextrins were decreasing with an increase in DE.
(2) EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), Mortensen, A., Aguilar, F., Crebelli, R., Di Domenico, A., Dusemund, B., Frutos, M.J., Galtier, P., Gott, D., Gundert‐Remy, U. and Lambré, C., 2017. Re‐evaluation of oxidised starch (E 1404), monostarch phosphate (E 1410), distarch phosphate (E 1412), phosphated distarch phosphate (E 1413), acetylated distarch phosphate (E 1414), acetylated starch (E 1420), acetylated distarch adipate (E 1422), hydroxypropyl starch (E 1440), hydroxypropyl distarch phosphate (E 1442), starch sodium octenyl succinate (E 1450), acetylated oxidised starch (E 1451) and starch aluminium octenyl succinate (E 1452) as food additives. EFSA Journal, 15(10), p.e04911.
Abstract. Following a request from the European Commission, the EFSA Panel on Food Additives and Nutrient sources added to Food (ANS) was asked to deliver a scientific opinion on the re-evaluation of 12 modified starches (E 1404, E 1410, E 1412, E 1413, E 1414, E 1420, E 1422, E 1440, E 1442, E 1450, E 1451 and E 1452) authorised as food additives in the EU in accordance with Regulation (EC) No 1333/2008 and previously evaluated by JECFA and the SCF. Both committees allocated an acceptable daily intake (ADI) ‘not specified’. In humans, modified starches are not absorbed intact but significantly hydrolysed by intestinal enzymes and then fermented by the intestinal microbiota. Using the read-across approach, the Panel considered that adequate data on short- and long-term toxicity and carcinogenicity, and reproductive toxicity are available. Based on in silico analyses, modified starches are considered not to be of genotoxic concern. No treatment-related effects relevant for human risk assessment were observed in rats fed very high levels of modified starches (up to 31,000 mg/kg body weight (bw) per day). Modified starches (e.g. E 1450) were well tolerated in humans up to a single dose of 25,000 mg/person. Following the conceptual framework for the risk assessment of certain food additives, the Panel concluded that there is no safety concern for the use of modified starches as food additives at the reported uses and use levels for the general population and that there is no need for a numerical ADI. The combined exposure to E 1404–E 1451 at the 95th percentile of the refined (brand-loyal) exposure assessment scenario for the general population was up to 3,053 mg/kg bw per day. Exposure to E 1452 for food supplement consumers only at the 95th percentile was up to 22.1 mg/kg bw per day.
| Evaluate |
