| "Descrizione" by Frank123 (11988 pt) | 2026-Jan-07 09:10 |
Review Consensus: 10 Rating: 10 Number of users: 1
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
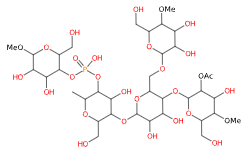
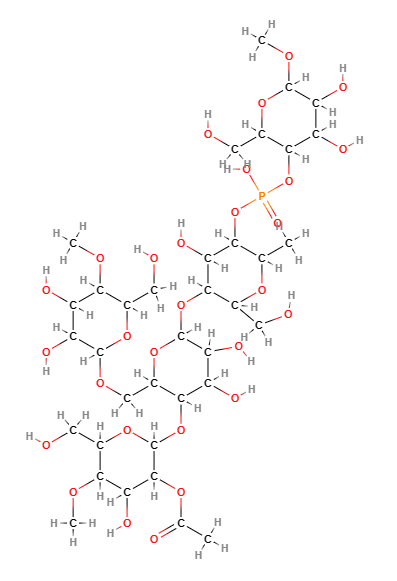
Acetylated distarch phosphate: properties, uses, pros and cons
Acetylated distarch phosphate is a modified starch produced by esterification of phosphorus oxychloride or sodium trimetaphosphate with an edible starch (rice or maize) and with esterification of vinyl acetate or acetic anhydride.
Breakdown of the name and function of the components
- Acetylated - Indicates that starch molecules have been modified with acetyl groups.
- Distarch - Refers to a modified starch.
- Phosphate - Indicates the presence of phosphate groups as part of the modification.
Description and function of the raw materials used in production
- Starch - A polysaccharide extracted mainly from cereals and tubers.
- Acetic anhydride - Used to introduce acetyl groups into starch.
- Phosphate - Used to introduce phosphate groups into starch.
Summary of its industrial synthesis process step by step
- Starch extraction - Starch is extracted from plants like corn, potatoes, or wheat.
- Acetylation - The extracted starch is treated with acetic anhydride to introduce acetyl groups.
- Phosphation - The acetylated starch is then treated with a phosphating agent to introduce phosphate groups.
- Purification - The modified starch is purified to remove any unreacted reagents or impurities.
- Drying - The product is dried to remove residual moisture.
It appears as a white powder or white granules.

What it is used for and where
Food
Ingredient listed in the European food additives list as E1414 as a stabiliser, thickener to provide consistency and preservation even in foods with high acidity. It is used to achieve a better quality in sterilised food products with a low pH value. Useful in the production of maltodextrins, it is however rather susceptible to enzymatic degradation (1).
Safety
The EFSA Panel on Food Additives and Nutrient Sources Added to Food considers that there is no safety concern for the use of modified starches as food additives at the uses and use levels declared for the general population and that a numerical ADI is not necessary (2).
 |  |
 | 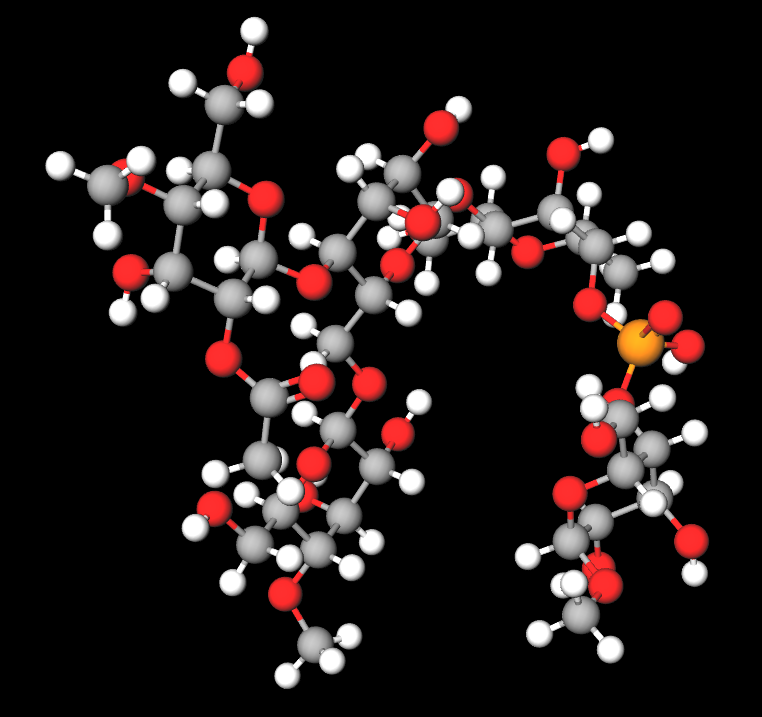 |
- Molecular Formula C36H63O29P
- Molecular Weight 990.8
- CAS 68130-14-3
- UNII
- EC Number 813-662-8
References_____________________________________________________________________
(1) Pycia, K., Juszczak, L., Gałkowska, D., Socha, R. and Jaworska, G., 2017. Maltodextrins from chemically modified starches. Production and characteristics. Starch‐Stärke, 69(5-6), p.1600199.
Abstract. The aim of this work was to evaluate whether the type of chemically modified starch (cross-linked and/or stabilised) has any effect on its susceptibility to α-amylase and on the physicochemical properties of the resulting maltodextrins of dextrose equivalent (DE) of 6, 11 and 16. The duration of the enzymatic hydrolysis of starch was established experimentally on the basis of a course of individual starch hydrolysis. The type of chemically modified starch was found to have a significant effect on its susceptibility to enzymatic hydrolysis. Native potato starch was more easily hydrolysed than acetylated starch and acetylated distarch adipate. The types of starch most susceptible to enzymatic degradation were distarch phosphate and acetylated distarch phosphate. The results of a chromatographic analysis revealed that the maltodextrins of DE 6 differed from one another significantly in terms of the amounts of each of the DP1–DP5 fractions. The number average molecular weight and the weight average molecular weight of the maltodextrins were decreasing with an increase in DE.
(2) EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), Mortensen, A., Aguilar, F., Crebelli, R., Di Domenico, A., Dusemund, B., Frutos, M.J., Galtier, P., Gott, D., Gundert‐Remy, U. and Lambré, C., 2017. Re‐evaluation of oxidised starch (E 1404), monostarch phosphate (E 1410), distarch phosphate (E 1412), phosphated distarch phosphate (E 1413), acetylated distarch phosphate (E 1414), acetylated starch (E 1420), acetylated distarch adipate (E 1422), hydroxypropyl starch (E 1440), hydroxypropyl distarch phosphate (E 1442), starch sodium octenyl succinate (E 1450), acetylated oxidised starch (E 1451) and starch aluminium octenyl succinate (E 1452) as food additives. EFSA Journal, 15(10), p.e04911.
Abstract. Following a request from the European Commission, the EFSA Panel on Food Additives and Nutrient sources added to Food (ANS) was asked to deliver a scientific opinion on the re-evaluation of 12 modified starches (E 1404, E 1410, E 1412, E 1413, E 1414, E 1420, E 1422, E 1440, E 1442, E 1450, E 1451 and E 1452) authorised as food additives in the EU in accordance with Regulation (EC) No 1333/2008 and previously evaluated by JECFA and the SCF. Both committees allocated an acceptable daily intake (ADI) ‘not specified’. In humans, modified starches are not absorbed intact but significantly hydrolysed by intestinal enzymes and then fermented by the intestinal microbiota. Using the read-across approach, the Panel considered that adequate data on short- and long-term toxicity and carcinogenicity, and reproductive toxicity are available. Based on in silico analyses, modified starches are considered not to be of genotoxic concern. No treatment-related effects relevant for human risk assessment were observed in rats fed very high levels of modified starches (up to 31,000 mg/kg body weight (bw) per day). Modified starches (e.g. E 1450) were well tolerated in humans up to a single dose of 25,000 mg/person. Following the conceptual framework for the risk assessment of certain food additives, the Panel concluded that there is no safety concern for the use of modified starches as food additives at the reported uses and use levels for the general population and that there is no need for a numerical ADI. The combined exposure to E 1404–E 1451 at the 95th percentile of the refined (brand-loyal) exposure assessment scenario for the general population was up to 3,053 mg/kg bw per day. Exposure to E 1452 for food supplement consumers only at the 95th percentile was up to 22.1 mg/kg bw per day.
| Evaluate |
