| "Descrizione" about Sodium Propyl Parahydroxybenzoate Review Consensus 10 by FRanier (9960 pt) | 2024-Oct-05 12:23 |
| Read the full Tiiip | (Send your comment) |
Propyl sodium parahydroxybenzoate, commonly known as sodium propylparaben, is a sodium salt of propylparaben, which belongs to the paraben family. It is used as a preservative in cosmetics, personal care products, and pharmaceuticals due to its antimicrobial properties, particularly its ability to prevent the growth of mold, yeast, and bacteria. This ensures that products maintain their quality and shelf life by preventing microbial contamination.
Chemical Composition and Structure
Propyl sodium parahydroxybenzoate is a paraben ester formed by the reaction of parahydroxybenzoic acid with propanol, then neutralized with sodium hydroxide. Its chemical structure consists of a benzoic acid core with a propyl group attached, which contributes to its antimicrobial efficacy. The sodium salt form improves its solubility in water, making it suitable for use in aqueous formulations.
Physical Properties
Propyl sodium parahydroxybenzoate typically appears as a white crystalline powder. It is water-soluble, which makes it ideal for use in water-based formulations. It is stable over a wide range of pH levels and temperatures, providing long-lasting preservative effects in a variety of products.
The name describes the structure of the molecule:
- Sodium indicates the presence of a sodium ion. Sodium is an alkali metal, and in this context, it's used to form a salt with parahydroxybenzoic acid, making the molecule water-soluble.
- Propyl indicates the propyl group, an alkyl chain with three carbon atoms. It's the lipophilic (or hydrophobic) component of the molecule, meaning it has a preference for oily or fatty substances.
- Parahydroxybenzoate is the main part of the molecule and indicates that it's a derivative of parahydroxybenzoic acid, a type of benzoic acid that has a hydroxy group in the para position on the benzene ring.
Raw Materials Used in Production.
Propyl parahydroxybenzoic acid is derived from benzoic acid, which can be sourced from benzoin or chemically synthesized.
Step-by-step Summary of Industrial Production Process.
- Synthesis of propyl parahydroxybenzoic acid. Benzoic acid is reacted with propyl alcohol to yield propyl parahydroxybenzoate ester.
- Neutralization. The ester is then neutralized with sodium hydroxide (NaOH) to form the salt, Sodium Propyl Parahydroxybenzoate.
- Purification. The product is purified through various methods such as filtration and crystallization to remove impurities.
Form and Color.
It appears as a white or almost white crystalline powder.
What it is for and where
Cosmetics
It is a restricted ingredient as V/12 a Relevant Item in the Annexes of the European Cosmetics Regulation 1223/2009. Substance or ingredient reported: Butyl 4-hydroxybenzoate and its salts Propyl 4-hydroxybenzoate and its salts
- Maximum concentration in ready for use preparation - 0,14% (as acid) for the sum of the individual concentrations - 0,8% (as acid) for mixtures of substances mentioned in entry 12 and 12a, here the sum of the individual concentrations of butyl and propylparaben and their salts does not exceed 0,14%
- Other Not to be used in leave-on products designed for application on the nappy area of children under three years of age.
- Wording of conditions of use and warnings For leave-on products designed for children under three years of age: "Do not use on the nappy area"
Preservative. Any product containing organic, inorganic compounds, water, needs to be preserved from microbial contamination. Preservatives act against the development of harmful microorganisms and against oxidation of the product.
Commercial Applications
Cosmetics Industry. Sodium Propylparaben is a preservative widely used in cosmetics and skincare products. It helps prevent the growth of microorganisms, thus extending product shelf life.
Food Industry. It's also utilized as a preservative in certain food products to prevent the growth of bacteria, molds, and yeasts.
Medicine. It may be used in some pharmaceutical formulations as a preservative.
Safety
11-6-2019 I had written to the European Commission's Directorate for Health and Food Safety (DG SANTE) reiterating doubts about the safety of parabens and E11 titanium dioxide. Finally, also from this body came the answer that clarifies all doubts:
"Regarding the use of methyl- and propylparaben as excipients in oral medicinal products for human use, I would advise you to look at the information provided by the European Medicines Agency at https://www.ema.europa.eu/en/use-methyl-propylparaben-excipients-human-medicinal-products-oral-use This discussion paper deals with methyl- and propylparaben, as these are the parabens predominantly used in oral pharmaceutical formulations. The focus of this paper is on possible endocrine disrupting effects in humans.
Parabens are components discussed and on whose safety many doubts have been raised, especially for the damage they would produce to the aquatic environment where they are discharged after use (1).
Parabens can contribute to obesity (2).
Parabens are preservative chemical compounds that have been the subject of attention in the scientific literature as possible endocrine disruptors (particularly propylparaben and butylparaben), i.e. with the possibility of damaging the hormone-producing glands in our bodies, particularly in the breasts. The 2004 study by Darbre et al. showed that parabens remain in our bodies as intact esters (2). Following this study, some of the scientific literature in 2005 and 2006 cast doubt on Darbre's conclusions and claimed they were limited. However, both the US FDA and the European SCCP authorised in 2006 the use of a single paraben in cosmetic products at a concentration of 0.4% and the use of total parabens at a concentration of 0.8%. However, there is no shortage of studies that consider the restrictions unnecessary: M. G. Kirchhof et al. in 2013 found that parabens are among the safest and most well-tolerated preservatives and that current data do not support drastic regulations or personal exposure restrictions. Darbre in 2014 published a further study in which he showed how parabens can cause DNA damage (3).
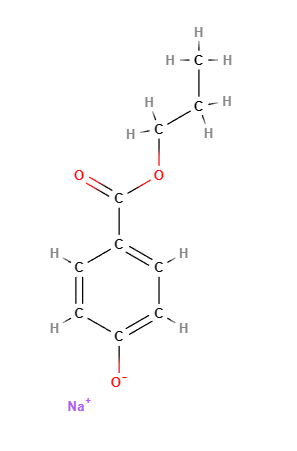 | 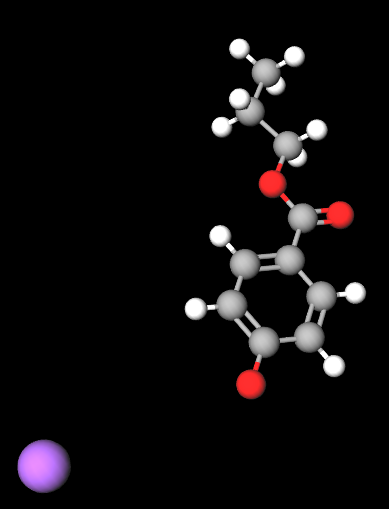 |
- Molecular Formula C10H11NaO3
- Molecular Weight 202.185 g/mol
- CAS 35285-69-9
- UNII 625NNB0G9N
- DTXSID3042348
- Nikkaji J42.046H
Synonyms :
sodium propyl p-hydroxybenzoate
Benzoic acid, 4-hydroxy-, propyl ester, sodium salt
Sodium 4-propoxycarbonylphenoxide
Sodium propylparaben
Propylparaben sodium
EINECS 252-488-1
sodium;4-propoxycarbonylphenolate
Sodium Propyl Parahydroxybenzoate
sodium;4-propoxycarbonylphenolate
Sodium propyl hydroxybenzoate
Sodium Propyl Parahydroxybenzoate
References___________________________________________________________________
(1) Terasaki M, Abe R, Makino M, Tatarazako N. Chronic toxicity of parabens and their chlorinated by-products in Ceriodaphnia dubia. Environ Toxicol. 2015 May-Jun;30(6):664-73. doi: 10.1002/tox.21944. Epub 2013 Dec 27. PMID: 24376163.
Popa DS, Bolfa P, Kiss B, Vlase L, Păltinean R, Pop A, Cătoi C, Crişan G, Loghin F. Influence of Genista tinctoria L. or methylparaben on subchronic toxicity of bisphenol A in rats. Biomed Environ Sci. 2014 Feb;27(2):85-96. doi: 10.3967/bes2014.021.
(2) Hu P, Chen X, Whitener RJ, Boder ET, Jones JO, Porollo A, Chen J, Zhao L. Effects of parabens on adipocyte differentiation. Toxicol Sci. 2013 Jan;131(1):56-70. doi: 10.1093/toxsci/kfs262.
Abstract. Parabens are a group of alkyl esters of p-hydroxybenzoic acid that include methylparaben, ethylparaben, propylparaben, butylparaben, and benzylparaben. Paraben esters and their salts are widely used as preservatives in cosmetics, toiletries, food, and pharmaceuticals. Humans are exposed to parabens through the use of such products from dermal contact, ingestion, and inhalation. However, research on the effects of parabens on health is limited, and the effects of parabens on adipogenesis have not been systematically studied. Here, we report that (1) parabens promote adipogenesis (or adipocyte differentiation) in murine 3T3-L1 cells, as revealed by adipocyte morphology, lipid accumulation, and mRNA expression of adipocyte-specific markers; (2) the adipogenic potency of parabens is increased with increasing length of the linear alkyl chain in the following potency ranking order: methyl- < ethyl- < propyl- < butylparaben. The extension of the linear alkyl chain with an aromatic ring in benzylparaben further augments the adipogenic ability, whereas 4-hydroxybenzoic acid, the common metabolite of all parabens, and the structurally related benzoic acid (without the OH group) are inactive in promoting 3T3-L1 adipocyte differentiation; (3) parabens activate glucocorticoid receptor and/or peroxisome proliferator-activated receptor γ in 3T3-L1 preadipocytes; however, no direct binding to, or modulation of, the ligand binding domain of the glucocorticoid receptor by parabens was detected by glucocorticoid receptor competitor assays; and lastly, (4) parabens, butyl- and benzylparaben in particular, also promote adipose conversion of human adipose-derived multipotent stromal cells. Our results suggest that parabens may contribute to obesity epidemic, and the role of parabens in adipogenesis in vivo needs to be examined further.
(3) Darbre PD, Harvey PW. Parabens can enable hallmarks and characteristics of cancer in human breast epithelial cells: a review of the literature with reference to new exposure data and regulatory status. J Appl Toxicol. 2014 Sep;34(9):925-38. doi: 10.1002/jat.3027.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Allura Red AC Review Consensus 10 by FRanier (9960 pt) | 2023-Jun-25 19:35 |
| Read the full Tiiip | (Send your comment) |
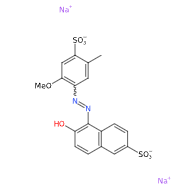
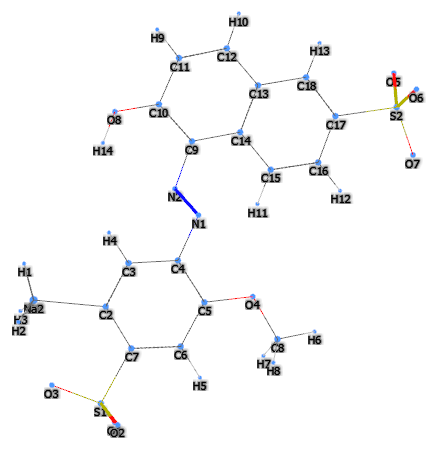
FD&C Red 40 is a synthetic chemical, azo derivative, water-soluble dye, referred to by many synonyms, of which the most common is CI 16035, Allura Red 40 or Allura Red AC and is among the most widely used dyes in the world.
The name describes the structure of the molecule:
- FD&C. This stands for Food, Drug, and Cosmetic. This prefix is used by the U.S. Food and Drug Administration (FDA) to denote dyes that are approved for use in foods, drugs, and cosmetics.
- Red 40. This is the specific name of the dye. Red 40, also known as Allura Red, is a red azo dye.
It occurs as a fine red powder. Soluble in water and ethanol.

The synthesis process takes place in different steps:
- Preparation. The synthesis of FD&C Red 40 begins with the preparation of the necessary raw materials. This typically includes naphthalene, which is a polycyclic aromatic hydrocarbon, and other chemicals.
- Sulphonation. Naphthalene undergoes a process called sulfonation, in which a group of sulfuric acid is added to the naphthalene molecule.
- Diazotization and coupling. Naphthalene sulfonate and diazotized, a procedure that involves replacing an amino group with a diazo group. This is followed by a coupling reaction, where diazo is combined with another molecule to form the azo dye.
- Purification. This is the final stage of the synthesis process and involves the removal of any raw materials and by-products not reacted by the reaction mixture to obtain the pure FD&C Red 40.
It occurs as a fine red powder. Soluble in water and ethanol.

It contains the azo group, which is the largest group of colours and accounts for more than half of global colour production. In food it is labelled as E129 on the European Food Additives List and is subject to an Acceptable Daily Intake of 0.7 mg/kg body weight (1).
Industrial dyes, which are used to colour food, drink, meat, sweets, hair dyes, face and body care cosmetics, fall into two categories:
- natural compounds or additives derived from the natural ingredient (i)
- synthetic chemicals (ii)
Safety
Some countries have regulated and, in some cases, banned the use of Allura red in food and beverages. It's approved for use in the EU, US, and many other countries, although there have been concerns about its potential to cause allergic reactions and its safety has been the subject of debate.
There is no consensus in the scientific literature that Allura Red is genotoxic. In vivo tests on laboratory animals have provided conflicting results (2).
Optimal typical characteristics of the commercial product Allura Red
| Appearance | Fine red powder |
| Boiling Point | |
| Melting Point | 300ºC |
| PSA | 185.34000 |
| LogP | 5.24760 |
| Arsenic | ≤ 0.0001% |
| Insoluble matter in water | ≤ 0.2% |
| Lead | ≤ 0.001% |
| Heavy metal (calculated by Pb) | ≤ 0.002% |
| Ether extraction | 0.2% max |
| Dye intermediates | 0.5% max |
| Loss on drying at 135°C, chlorides, sulphates | 13% max |
| Mercury | <1 ppm |
 |  |
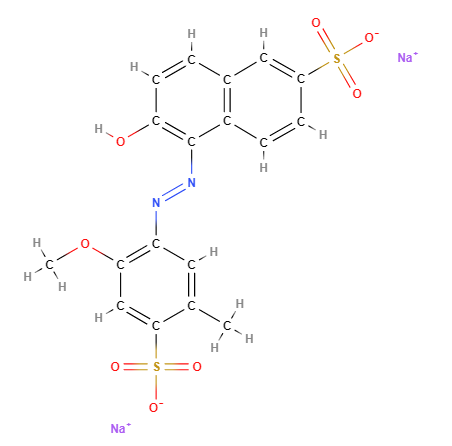 | 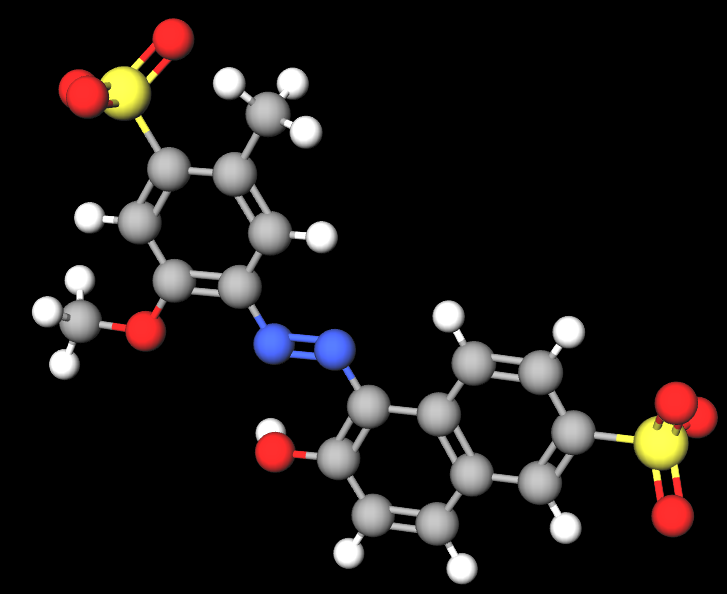 |
- Molecular Formula : C18H14N2Na2O8S2
- Molecular Weight : 496.42
- Exact mass 495.998688
- CAS : 25956-17-6
- UNII WZB9127XOA
- EC Number 247-368-0
- DSSTox Substance ID DTXSID4024436
- MDL number MFCD00059526
- PubChem Substance ID 24869338
- InChI=1S/C18H16N2O8S2.2Na/c1-10-7-14(16(28-2)9-17(10)30(25,26)27)19-20-18-13-5-4-12(29(22,23)24)8-11(13)3-6-15(18)21;;/h3-9,21H,1-2H3,(H,22,23,24)(H,25,26,27);;/q;2*+1/p-2
- InChl Key CEZCCHQBSQPRMU-UHFFFAOYSA-L
- SMILES CC1=CC(=C(C=C1S(=O)(=O)[O-])OC)N=NC2=C(C=CC3=C2C=CC(=C3)S(=O)(=O)[O-])O.[Na+].[Na+]
- IUPAC disodium;6-hydroxy-5-[(2-methoxy-5-methyl-4-sulfonatophenyl)diazenyl]naphthalene-2-sulfonate
- ChEBI 172687
- NACRES NA.47
Synonyms :
- Allura Red AC
- Allura Red 40
- FDC Red 40
- Red No. 40
- Allura red AC dye
- C.I. Food Red 17
- Food red 17
- Food Red No. 40
- FD&C Red No. 40
- Disodium 6-hydroxy-5-((6-methoxy-4-sulfo-m-tolyl)azo)-2-naphthalenesulfonate
- 2-Naphthalenesulfonic acid, 6-hydroxy-5-((6-methoxy-4-sulfo-m-tolyl)azo)-, disodium salt
- Disodium 6-hydroxy-5-((2-methoxy-5-methyl-4-sulfophenyl)azo)-2-naphthalenesulfonate
References________________________________________________________________________
(1) Rovina K, Siddiquee S, Shaarani SM. Extraction, Analytical and Advanced Methods for Detection of Allura Red AC (E129) in Food and Beverages Products. Front Microbiol. 2016 May 27;7:798. doi: 10.3389/fmicb.2016.00798.
(2) Tsuda S, Murakami M, Matsusaka N, Kano K, Taniguchi K, Sasaki YF. DNA damage induced by red food dyes orally administered to pregnant and male mice. Toxicol Sci. 2001 May;61(1):92-9. doi: 10.1093/toxsci/61.1.92.
Ferrer Amate C, Unterluggauer H, Fischer RJ, Fernández-Alba AR, Masselter S. Development and validation of a LC-MS/MS method for the simultaneous determination of aflatoxins, dyes and pesticides in spices. Anal Bioanal Chem. 2010 May;397(1):93-107. doi: 10.1007/s00216-010-3526-x.
Honma M. Evaluation of the in vivo genotoxicity of Allura Red AC (Food Red No. 40). Food Chem Toxicol. 2015 Oct;84:270-5. doi: 10.1016/j.fct.2015.09.007.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Description" about Invert Syrup Review Consensus 10 by FRanier (9960 pt) | 2024-Apr-08 11:00 |
| Read the full Tiiip | (Send your comment) |
Invert syrup is a commercial mixture composed of Sucrose, Glucose, and Fructose. Invert sugar is a sweetener made by breaking sucrose, or table sugar, into its two component sugars, glucose and fructose, through a process called hydrolysis. This produces a syrup that is sweeter than sucrose, with a softer mouthfeel and improved preservative qualities due to its high solubility and moisture retention properties. Invert sugar is commonly used in the food industry to sweeten a wide range of products, including candies, baked goods, and beverages, as well as in ice cream to prevent crystallization and improve texture.
The composition usually is:
- Glucose 39%.
- Fructose 36% (1)
- Water 20%
- Sucrose 5%
Nutritional Profile (per 100 grams):
- Calories. About 310 kcal, similar to that of regular sugar.
- Carbohydrates. About 76 grams, mainly composed of equal parts glucose and fructose.
- Protein. Negligible protein content.
- Fats. No fat content.
- Fiber. No fiber content.
- Sodium. Minimum sodium content.
The sweetening power of this syrup is much higher than that of simple sugar.
- Composition. Inverted sugar syrup is composed of glucose and fructose in nearly equal proportions, resulting from the splitting of sucrose. The presence of these two simple sugars gives it unique characteristics compared to pure table sugar.
- Sweetening Properties. The sweetness of inverted sugar syrup is greater than that of pure sucrose, making it particularly useful in recipes requiring a high sweetening power without adding large amounts of sugar.
- Preservation and Stability. It improves the shelf life of food products by reducing sugar crystallization and increasing stability in acidic solutions, such as soft drinks. This makes it ideal for confectionery, ice creams, and baked goods.
- Moisture. It attracts and retains moisture better than sucrose, helping to keep baked products soft and fresh for longer.
Industrial Production Process
- Preparation. Sucrose, primarily extracted from sugar cane or sugar beet, is dissolved in water to create a concentrated solution.
- Acidification. The sucrose solution is acidified by adding an acid, such as citric acid or tartaric acid, to lower the pH. This step facilitates the hydrolysis of sucrose into glucose and fructose.
- Hydrolysis. Acidification initiates the hydrolysis of sucrose. Alternatively, hydrolysis can be accelerated through the use of specific enzymes, like invertase, which efficiently cleave sucrose at controlled temperatures.
- Neutralization. If necessary, the solution is neutralized by adding a base, such as sodium bicarbonate, to correct the acidity and stabilize the pH of the final product.
- Concentration. The inverted sugar solution is concentrated by vacuum evaporation to achieve the desired syrup consistency.
- Quality Control. The inverted sugar syrup undergoes quality control checks to verify composition, purity, density, and the absence of contaminants, ensuring it meets the required specifications.
Safety
Impact on Metabolism. Although inverted sugar syrup is metabolized similarly to other simple sugars, its high fructose content can contribute to the same health risks associated with excessive fructose consumption, such as weight gain and glucose metabolism disorder.
Fructose is a naturally occurring sugar found in fruits, vegetables, and honey. Fructose is another component with a harmless and inviting name, but whose excessive consumption can create health risks. This ingredient is often included in desserts, beverages "sugar-free" food products, etc. to increase the sweet taste and we find it in glucose-fructose syrups, fructose syrups, invert sugar and even pure sugar.
Excessive consumption of invert sugar can induce metabolic alterations to glucose and DNA (2).
Safety
During production, there are points of contamination by microorganisms that can change the properties of sugar and reduce its shelf life (3).
References_____________________________________________________________________
(1) ASHARE R, MOORE R, ELLISON EH. Utilization of glucose, fructose and invert sugar; comparison in diseases of the liver and pancreas. AMA Arch Surg. 1955 Mar;70(3):428-35. doi: 10.1001/archsurg.1955.01270090106024. PMID: 14349507.
(2) Molz P, Molz WA, Dallemole DR, Santos LFS, Salvador M, Cruz DB, PrÁ D, Franke SIR. Invert sugar induces glucose intolerance but does not cause injury to the pancreas nor permanent DNA damage in rats. An Acad Bras Cienc. 2020;92(2):e20191423. doi: 10.1590/0001-3765202020191423. Epub 2020 Jul 20. PMID: 32696841.
Abstract. The high consumption of sugars is linked to the intermediate hyperglycemia and impaired glucose tolerance associated with obesity, inducing the prediabetes. However, the consequences of excessive invert sugar intake on glucose metabolism and genomic stability were poorly studied. The aim of this study was to evaluate the effects of invert sugar overload (32%) in rats, analyzing changes in obesity, glucose tolerance, pancreatic/hepatic histology and primary and permanent DNA damage. After 17 weeks, the rats became obese and had an excessive abdominal fat, as well as presented impaired glucose tolerance, caused by higher sugar caloric intake. Primary DNA damage, evaluated by the comet assay, was increased in the blood, however not in the pancreas. No protein carbonylation was seen in serum. Moreover, no increase in permanent DNA damage was seen in the bone marrow, evaluated using the micronucleus test. Some rats presented liver steatosis and that the pancreatic islets were enlarged, but not significantly. In this study, invert sugar altered the glucose metabolism and induced primary DNA damage in blood, but did not cause significant damage to the pancreas or liver, and neither changes in the levels of oxidative stress or permanent DNA damage.
(3) Podadera, P. and Sabato, S.F., 2007, July. Radiation effect on sucrose content of inverted sugar. In International Nuclear Atlantic Conference. INAC.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Inverted sugar Review Consensus 10 by FRanier (9960 pt) | 2025-Nov-23 18:01 |
| Read the full Tiiip | (Send your comment) |
Inverted sugar is a commercial mixture composed of Sucrose, Glucose, and Fructose. Invert sugar is a sweetener made by breaking sucrose, or table sugar, into its two component sugars, glucose and fructose, through a process called hydrolysis. This produces a syrup that is sweeter than sucrose, with a softer mouthfeel and improved preservative qualities due to its high solubility and moisture retention properties. Invert sugar is commonly used in the food industry to sweeten a wide range of products, including candies, baked goods, and beverages, as well as in ice cream to prevent crystallization and improve texture. However, like all sugars, it should be consumed in moderation as part of a balanced diet due to the potential health risks associated with high sugar intake, including obesity, type 2 diabetes, and heart disease.
The composition usually is :
- Glucose 39%.
- Fructose 36% (1)
- Water 20%
- Sucrose 5%

Industrial Production Process
- Preparation of Sugar Solution. The production of inverted sugar starts with preparing an aqueous solution of sucrose (common table sugar). This solution is slightly heated to facilitate the subsequent inversion reaction.
- Acidification. An acid, such as citric acid or tartaric acid, is added to the sucrose solution to lower the pH. The acidity is necessary to catalyze the cleavage of sucrose into glucose and fructose, the components of inverted sugar.
- Heating. The solution is heated to a controlled temperature, generally between 50°C and 60°C, for a set period to promote the inversion reaction.
- Neutralization. After the inversion reaction is complete, the solution is neutralized by adding a base, such as sodium bicarbonate, to bring the pH to a neutral level and stop the reaction.
- Cooling and Filtration. The inverted sugar solution is cooled to room temperature and filtered to remove any impurities or sediments.
- Concentration. If necessary, the solution can be further concentrated by vacuum evaporation to achieve the desired consistency.
Physical properties
Appearance: clear, viscous liquid, from colourless to pale yellow.
Density: about 1.30–1.40 g/mL (depending on Brix and temperature).
Viscosity: high; increases with solids content and decreases with temperature.
Solubility: fully miscible in water.
pH: typically 4–6 (depending on process and neutralisation).
Sensory and technological properties
Sweetness: slightly higher than sucrose due to fructose, which has greater sweetening power.
Strong humectant capacity, helping retain moisture and softness in baked goods and chewy confectionery.
Excellent anti-crystallising behaviour; inhibits sucrose crystallisation in syrups, fondants, ice cream and candies.
Improves freeze–thaw stability in ice cream and sorbets.
Helps stabilise flavour compounds in liquid and semi-liquid systems.
Can contribute to lower water activity in some recipes, enhancing shelf-life.
Food applications
Bakery & pastry: cakes, sponge cakes, festive cakes, soft cookies, fillings, glazes, icings.
Ice cream & frozen desserts: gelato bases, sorbets, ice cream, where it helps texture, sweetness and anti-crystallisation.
Confectionery: soft candies, gums, fondants, fillings, jellies.
Beverages: flavored syrups, soft drinks, iced teas, functional and sports drinks.
Industrial baked goods: snack cakes, pastries, croissants, sweet rolls.
Other applications: dessert sauces, fruit preparations, specialised jams, toppings.
Nutrition & health
Invert sugar is a source of simple carbohydrates, with the same energy as sucrose on a gram-for-gram basis.
The presence of fructose increases perceived sweetness, but does not reduce calories unless overall sugar use is lowered.
Excessive intake of simple sugars may contribute to:
weight gain and excess caloric intake,
dental caries,
poorer glycaemic control in susceptible individuals.
Should be consumed in moderation, as part of a balanced diet, in line with recommendations on added sugars.
Portion note
In beverages: typically 5–10 g per serving (≈ 1–2 teaspoons).
In ice cream and pastry formulations: often 3–25% of the total recipe, depending on texture, sweetness and anti-crystallisation needs.
Allergens and intolerances
Invert sugar is not a major allergen.
Naturally gluten-free and lactose-free, unless cross-contamination occurs in shared facilities.
Its use should be evaluated in people with dietary restrictions on simple sugars (e.g., some metabolic conditions).
Storage & shelf-life
Store in tightly closed containers in a cool, dry place, away from direct heat and sunlight.
Recommended storage temperature: 15–25 °C.
Typical shelf-life: 12–24 months if unopened and properly stored.
Main risks:
fermentation if microbiologically contaminated or diluted;
browning from slow oxidation or mild caramelisation;
changes in viscosity over time with temperature fluctuations.
Safety & regulatory
Considered a standard food ingredient (derived from sucrose and water).
Must comply with:
purity and contaminant criteria for sugar;
potable water requirements;
applicable microbiological criteria.
Manufactured in accordance with GMP/HACCP, ensuring full traceability of raw materials and batches.
Labeling
May be declared as:
“invert sugar”,
“invert sugar syrup”.
In compound foods, it appears in the ingredient list in descending order of weight.
It may be highlighted for its technological role in ice cream, confectionery and soft baked products.
Troubleshooting
Unexpected crystallisation:
inversion level too low → optimise inversion process;
Brix not appropriate → adjust concentration and formulation.
Fermentation or gas formation in containers:
microbial contamination → improve hygiene, process controls and storage conditions.
Too dark colour / caramelised flavour:
excessive process temperature or long concentration times → optimise heating profile.
Off-spec viscosity:
Brix out of target → re-adjust solids;
large temperature variations → consider temperature impact during processing and use.
Sustainability & supply chain
Linked to the sugar supply chain (cane or beet):
water use and irrigation in agriculture;
soil management and agricultural inputs (fertilisers, crop protection products);
environmental impact of sugar mills and refineries.
Production of invert sugar syrup requires energy (heating, evaporation) and generates wash waters and effluents that should be treated, often monitored using BOD/COD indicators.
Organic and fair trade sugar sources can be used to produce more sustainable invert sugar syrups.
Main INCI functions (cosmetics)
(when used in cosmetic products, not as a flavour)
Humectant – helps retain moisture in skin and hair formulations.
Solvent – for water-soluble actives.
Viscosity modifier – contributes to texture and rheology of gels, serums and lotions.
May appear as “Invert Sugar”, “Hydrolyzed Sucrose” or similar names, depending on the grade and application.
Conclusion
Invert sugar is a highly versatile sweetening and functional ingredient, improving sweetness, moisture retention, softness and anti-crystallisation in a wide variety of foods, especially ice cream, baked goods and confectionery. While it does not offer caloric advantages over sucrose, it allows finer control of texture, stability and sensory quality. When produced and handled according to GMP/HACCP and good storage practices, it is a safe, stable and high-quality ingredient for both industrial and artisanal applications.
Mini-glossary
SFA – Saturated fatty acids: a class of fats associated with increased cardiovascular risk when consumed in excess; not relevant in invert sugar, which contains no fat.
MUFA – Monounsaturated fatty acids: another fat class; not applicable here.
PUFA – Polyunsaturated fatty acids: oxidation-prone fats; not present in this ingredient.
TFA – Trans fatty acids: a fat type associated with negative health effects; not present in invert sugar.
GMP/HACCP – Good Manufacturing Practices / Hazard Analysis and Critical Control Points, systems to ensure hygiene, quality and safety in food production.
BOD/COD – Biological / Chemical Oxygen Demand, indicators of the environmental impact of wastewater from industrial processes.
Brix – Measure of soluble solids (mainly sugars) in a solution, crucial for controlling concentration and viscosity of syrups and invert sugar.
Considerations
Sugar Content: While invert sugar may improve the texture and shelf life of foods, it is still a form of sugar and contributes to the total sugar intake.
Health Impacts: High consumption of sugars, including invert sugar, can lead to health issues such as weight gain and an increased risk of chronic diseases.
Use in Cooking: Invert sugar syrup can be used at home to make candies and baked goods or to sweeten drinks. It's particularly useful in recipes that require a smooth texture or extended shelf life.
The sweetening power of this sugar, sometimes also called syrup, is far superior to that of simple sugar. Therefore, the advice is to consume it moderately and not overdo it to avoid cardiovascular risks.
Excessive consumption of invert sugar can induce metabolic alterations to glucose and DNA (2).
Safety
During production, there are points of contamination by microorganisms that can change the properties of sugar and reduce shelf life (3).
References_____________________________________________________________________
(1) ASHARE R, MOORE R, ELLISON EH. Utilization of glucose, fructose and invert sugar; comparison in diseases of the liver and pancreas. AMA Arch Surg. 1955 Mar;70(3):428-35. doi: 10.1001/archsurg.1955.01270090106024. PMID: 14349507.
(2) Molz P, Molz WA, Dallemole DR, Santos LFS, Salvador M, Cruz DB, PrÁ D, Franke SIR. Invert sugar induces glucose intolerance but does not cause injury to the pancreas nor permanent DNA damage in rats. An Acad Bras Cienc. 2020;92(2):e20191423. doi: 10.1590/0001-3765202020191423. Epub 2020 Jul 20. PMID: 32696841.
Abstract. The high consumption of sugars is linked to the intermediate hyperglycemia and impaired glucose tolerance associated with obesity, inducing the prediabetes. However, the consequences of excessive invert sugar intake on glucose metabolism and genomic stability were poorly studied. The aim of this study was to evaluate the effects of invert sugar overload (32%) in rats, analyzing changes in obesity, glucose tolerance, pancreatic/hepatic histology and primary and permanent DNA damage. After 17 weeks, the rats became obese and had an excessive abdominal fat, as well as presented impaired glucose tolerance, caused by higher sugar caloric intake. Primary DNA damage, evaluated by the comet assay, was increased in the blood, however not in the pancreas. No protein carbonylation was seen in serum. Moreover, no increase in permanent DNA damage was seen in the bone marrow, evaluated using the micronucleus test. Some rats presented liver steatosis and that the pancreatic islets were enlarged, but not significantly. In this study, invert sugar altered the glucose metabolism and induced primary DNA damage in blood, but did not cause significant damage to the pancreas or liver, and neither changes in the levels of oxidative stress or permanent DNA damage.
(3) Podadera, P. and Sabato, S.F., 2007, July. Radiation effect on sucrose content of inverted sugar. In International Nuclear Atlantic Conference. INAC.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about FD&C Red 40 Review Consensus 10 by FRanier (9960 pt) | 2023-Mar-23 18:11 |
| Read the full Tiiip | (Send your comment) |
FD&C Red 40 is a synthetic chemical, azo derivative, water-soluble dye, referred to by many synonyms, of which the most common is Allura Red 40 or Allura Red AC and is among the most widely used dyes in the world.
It occurs as a fine red powder. Soluble in water and ethanol.

It contains the azo group, which is the largest group of colours and accounts for more than half of global colour production. In food it is labelled as E129 on the European Food Additives List and is subject to an Acceptable Daily Intake of 0.7 mg/kg body weight (1).
Industrial dyes, which are used to colour food, drink, meat, sweets, hair dyes, face and body care cosmetics, fall into two categories:
- natural compounds or additives derived from the natural ingredient (i)
- synthetic chemicals (ii)
Safety
Some countries have regulated and, in some cases, banned the use of Allura red in food and beverages. Currently
There is no consensus in the scientific literature that Allura Red is genotoxic. In vivo tests on laboratory animals have provided conflicting results (2).
Optimal typical characteristics of the commercial product Allura Red
| Appearance | Fine red powder |
| Boiling Point | |
| Melting Point | 300ºC |
| PSA | 185.34000 |
| LogP | 5.24760 |
| Arsenic | ≤ 0.0001% |
| Insoluble matter in water | ≤ 0.2% |
| Lead | ≤ 0.001% |
| Heavy metal (calculated by Pb) | ≤ 0.002% |
| Ether extraction | 0.2% max |
| Dye intermediates | 0.5% max |
| Loss on drying at 135°C, chlorides, sulphates | 13% max |
| Mercury | <1 ppm |
 |  |
 |  |
- Molecular Formula : C18H14N2Na2O8S2
- Molecular Weight : 496.42
- Exact mass 495.998688
- CAS : 25956-17-6
- UNII WZB9127XOA
- EC Number 247-368-0
- DSSTox Substance ID DTXSID4024436
- MDL number MFCD00059526
- PubChem Substance ID 24869338
- InChI=1S/C18H16N2O8S2.2Na/c1-10-7-14(16(28-2)9-17(10)30(25,26)27)19-20-18-13-5-4-12(29(22,23)24)8-11(13)3-6-15(18)21;;/h3-9,21H,1-2H3,(H,22,23,24)(H,25,26,27);;/q;2*+1/p-2
- InChl Key CEZCCHQBSQPRMU-UHFFFAOYSA-L
- SMILES CC1=CC(=C(C=C1S(=O)(=O)[O-])OC)N=NC2=C(C=CC3=C2C=CC(=C3)S(=O)(=O)[O-])O.[Na+].[Na+]
- IUPAC disodium;6-hydroxy-5-[(2-methoxy-5-methyl-4-sulfonatophenyl)diazenyl]naphthalene-2-sulfonate
- ChEBI 172687
- NACRES NA.47
Synonyms :
- Allura Red AC
- Allura Red 40
- FDC Red 40
- Red No. 40
- Allura red AC dye
- C.I. Food Red 17
- Food red 17
- Food Red No. 40
- FD&C Red No. 40
- Disodium 6-hydroxy-5-((6-methoxy-4-sulfo-m-tolyl)azo)-2-naphthalenesulfonate
- 2-Naphthalenesulfonic acid, 6-hydroxy-5-((6-methoxy-4-sulfo-m-tolyl)azo)-, disodium salt
- Disodium 6-hydroxy-5-((2-methoxy-5-methyl-4-sulfophenyl)azo)-2-naphthalenesulfonate
References________________________________________________________________________
(1) Rovina K, Siddiquee S, Shaarani SM. Extraction, Analytical and Advanced Methods for Detection of Allura Red AC (E129) in Food and Beverages Products. Front Microbiol. 2016 May 27;7:798. doi: 10.3389/fmicb.2016.00798.
(2) Tsuda S, Murakami M, Matsusaka N, Kano K, Taniguchi K, Sasaki YF. DNA damage induced by red food dyes orally administered to pregnant and male mice. Toxicol Sci. 2001 May;61(1):92-9. doi: 10.1093/toxsci/61.1.92.
Ferrer Amate C, Unterluggauer H, Fischer RJ, Fernández-Alba AR, Masselter S. Development and validation of a LC-MS/MS method for the simultaneous determination of aflatoxins, dyes and pesticides in spices. Anal Bioanal Chem. 2010 May;397(1):93-107. doi: 10.1007/s00216-010-3526-x.
Honma M. Evaluation of the in vivo genotoxicity of Allura Red AC (Food Red No. 40). Food Chem Toxicol. 2015 Oct;84:270-5. doi: 10.1016/j.fct.2015.09.007.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Sodium myreth sulfate studies" about Sodium myreth sulfate Review Consensus 8 by FRanier (9960 pt) | 2022-Apr-16 10:30 |
| Read the full Tiiip | (Send your comment) |
Compendium of the most significant studies with reference to properties, intake, effects.
McNamara PJ, Syverson RE, Milligan-Myhre K, Frolova O, Schroeder S, Kidder J, Hoang T, Proctor RA. Surfactants, aromatic and isoprenoid compounds, and fatty acid biosynthesis inhibitors suppress Staphylococcus aureus production of toxic shock syndrome toxin 1. Antimicrob Agents Chemother. 2009 May;53(5):1898-906. doi: 10.1128/AAC.01293-08.
Abstract. We report here on the TSST-1-suppressing activity of 34 compounds that are commonly used additives in the pharmaceutical, food, and perfume industries.
Robinson, V. C., Bergfeld, W. F., Belsito, D. V., Hill, R. A., Klaassen, C. D., Marks, J. G., ... & Andersen, F. A. (2010). Final report of the amended safety assessment of sodium laureth sulfate and related salts of sulfated ethoxylated alcohols. International journal of toxicology, 29(4_suppl), 151S-161S.
Abstract. The potential to produce irritation exists with these salts of sulfated ethoxylated alcohols, but in practice they are not regularly seen to be irritating because of the formulations in which they are used. These ingredients should be used only when they can be formulated to be nonirritating.
Bährle-Rapp, M. (2007). Sodium Myreth Sulfate. In Springer Lexikon Kosmetik und Körperpflege (pp. 514-514). Springer, Berlin, Heidelberg.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Sodium myreth sulfate Review Consensus 10 by FRanier (9960 pt) | 2023-Dec-08 10:45 |
| Read the full Tiiip | (Send your comment) |
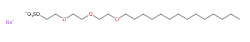
Sodium myreth sulfate is a chemical compound, an anionic surfactant, ethoxylated (reaction by polymerisation of ethylene oxide).
The name defines the structure of the molecule:
- Sodium (Na). Sodium is an alkaline metal, a chemical element with the symbol Na (from the Latin "Natrium") and the atomic number 11. Sodium is soft and silvery-white in color. It is highly reactive, especially with water, and is commonly found in salts and minerals.
- Myreth refers to a synthetic compound that is derived from miristyl alcohol, a fatty alcohol that is often used in cosmetics and personal care products.
- Sulfate refers to a salt or ester of sulfuric acid. Sulfates are often used in cosmetics and personal care products for their ability to create a foam, common in shampoos and body washes.
The synthesis process takes place in several stages:
- Ethoxylation. Myristyl alcohol, which is obtained from natural sources such as coconut oil or can be synthesized from myristic acid and made to react with ethylene oxide in a treatment called ethoxylation. The number of ethylene oxide units added varies in general, but for sodium myrtle sulfate, the "myreth" indicates that on average 10 units of ethylene oxide per molecule of Myristyl alcohol are added.
- Sulfation. Myristyl alcoholl ethoxylate is reacted with sulfur trioxide (SO3) to add a sulfate group, forming the corresponding sulfuric acid ester.
- Neutralization. The sulfuric acid ester is neutralized with sodium hydroxide (NaOH) to form Sodium Myreth Sulfate.
It typically appears as a clear to yellowish liquid at room temperature. It is water-soluble and is often used in aqueous solutions.

What it is used for and where
Cosmetics
Surfactant-foaming (1) emulsifying agent that facilitates amalgamation between the various ingredients. Emulsifiers have the property of directly influencing the stability, sensory properties and surface tension of sunscreens by modulating their filmometric performance.
INCI functions:
Cleansing agent. Ingredient that cleanses skin without exploiting the surface-active properties that produce a lowering of the surface tension of the stratum corneum.
Foaming. Its function is to introduce gas bubbles into the water for a purely aesthetic factor, which does not affect the cleaning process, but only satisfies the commercial aspect of the detergent by helping to spread the detergent. This helps in the commercial success of a cleansing formulation. Since sebum has an inhibiting action on the bubble, more foam is produced in the second shampoo. In practice, it creates many small bubbles of air or other gases within a small volume of liquid, changing the surface tension of the liquid.
Surfactant - Cleansing agent. Cosmetic products used to cleanse the skin utilise the surface-active action that produces a lowering of the surface tension of the stratum corneum, facilitating the removal of dirt and impurities.
Surfactant - Emulsifying agent. Emulsions are thermodynamically unstable and are used to soothe or soften the skin and emulsify, so they need a specific, stabilising ingredient. This ingredient forms a film, lowers the surface tension and makes two immiscible liquids miscible. A very important factor affecting the stability of the emulsion is the amount of the emulsifying agent. Emulsifiers have the property of reducing the oil/water or water/oil interfacial tension, improving the stability of the emulsion and also directly influencing the stability, sensory properties and surface tension of sunscreens by modulating the filmometric performance.
Use
- Shampoo 1%
- Shower 8%
- Hair dye 2,5%
Safety
After the ethoxylation procedure, scientific literature reports that it is not uncommon to find residues of ethylene oxide and 1,4-dioxane, chemical compounds that are considered carcinogenic, in the final product. It would therefore be necessary for the manufacturer to declare the purity of the ethoxylated chemical to be sure that the ingredient is harmless.
The CIR Expert Panel is of the opinion that Sodium myreth sulfate is safe as a cosmetic ingredient in the concentrations and uses established by cosmetic standards (2).
The most relevant studies on this ingredient have been selected with a summary of their contents:
| Appearance | Powder |
| PSA | 102.50000 |
| LogP | 5.29490 |
 |  |
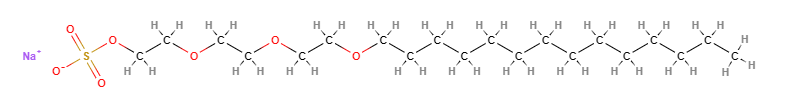 | 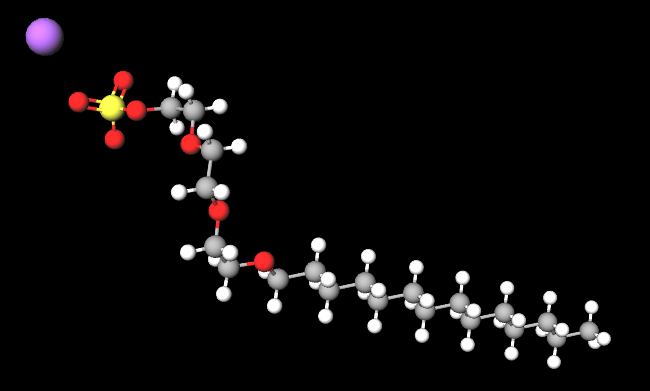 |
- Molecular Formula C20H41NaO7S
- Molecular Weight 448.6
- Exact Mass 448.24700
- CAS 25446-80-4
- UNII 2VLC033A4E
- EC Number 246-986-8
- DSSTox Substance ID DTXSID6067096
- IUPAC sodium;2-[2-(2-tetradecoxyethoxy)ethoxy]ethyl sulfate
- InChI=1S/C20H42O7S.Na/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-24-15-16-25-17-18-26-19-20-27-28(21,22)23;/h2-20H2,1H3,(H,21,22,23);/q;+1/p-1
- InChl Key MDSQKJDNWUMBQQ-UHFFFAOYSA-M
- SMILES CCCCCCCCCCCCCCOCCOCCOCCOS(=O)(=O)[O-].[Na+]
- MDL number
- PubChem Substance ID
- RTECS KM2931050
Synonyms:
- sodium 2-[2-[2-(tetradecyloxy)ethoxy]ethoxy]ethyl sulphate
- Polyethylene glycol (1-4) myristyl ether sulfate,sodium salt
- Ethanol,2-(2-(2-(tetradecyloxy)ethoxy)ethoxy)-,hydrogen sulfate,sodium salt
- 2-(2-(2-(Tetradecyloxy)ethoxy)ethoxy)ethanol hydrogen sulfate sodium salt
References_____________________________________________________________________
(1) Petrovska, L. S., Baranova, I. I., Bezpala, Y. O., & Kovalenko, S. M. (2017). The study of physicocochemical parameters of some detergents with the anionic nature.
Abstract. The physico-chemical parameters of etergents with the anionic nature, namely sodium myreth sulfate and sodium lauryl sarcosinate used most often when developing domestic foaming products for personal hygiene, at the cosmetic enterprises of Ukraine have been studied and analyzed. The indica-tors of the foaming ability (foam number, foam stability) of the test samples at different pH values, namely at the neutral pH (5.5 -6.0) and at the acid рН (3.5-4.0), have been determined and studied. The study of the foam structure at the pH intervals under research has been also conducted using the method of microphotography. According to the results of the study, the conclusion has been made that different pH values do not affect the foaming ability; however, they affect the foam structure.
(2) Final Report on the Safety Assessment of Sodium Myreth Sulfate. Journal of the American College of Toxicology. 1992;11(1):157-163. doi:10.3109/10915819209141996
Abstract. Sodium Myreth Sulfate is the sodium salt of sulfated, ethoxylated myristyl alcohol which is used as a surfactant and cleansing agent in cosmetics at concentrations ranging from > 1.0–5.0% to > 50.0%. A formulation containing 7.0% Sodium Myreth Sulfate was shown to be an ocular irritant in experimental animals and in some human test subjects. These irritant effects were similar to those previously reported for the chemically similar compound Sodium Laureth Sulfate which was shown to be safe for use in cosmetics. The report summarizes the safety test data on Sodium Laureth Sulfate. Based upon the combined data cited in the report on both cosmetic ingredients, it is concluded that Sodium Myreth Sulfate is safe as a cosmetic ingredient in the present practices of use and concentration.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips in __Italiano (32)
