App "Tiiips Ingredients Pro", Live Healthier!


App "Tiiips Ingredients Pro", Live Healthier!


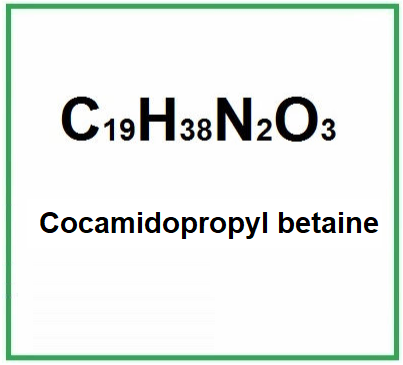
![]() Cocamidopropyl betaine
Cocamidopropyl betaine
Rating : 6
| Evaluation | N. Experts | Evaluation | N. Experts |
|---|---|---|---|
| 1 | 6 | ||
| 2 | 7 | ||
| 3 | 8 | ||
| 4 | 9 | ||
| 5 | 10 |
Cons:
Specific allergy (1)10 pts from Ark90
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.Evaluate | Where is this found? |
| "Cocamidopropyl betaine studies" about Cocamidopropyl betaine Review Consensus 7 by Al222 (18001 pt) | 2021-Dec-25 19:19 |
| Read the full Tiiip | (Send your comment) |
Irritation from surfactants in detergents is a rather frequent reaction in the use of cosmetics and sensitisation to the product is also possible. Cocamidopropyl betaine in the Italian standard (Società Italiana di Dermatologia Allergologica, Professionale e Ambientale [SIDAPA]) gave five positive reactions among 105 patients in a scientific test (1).
Other studies have shown allergies, but to a lesser extent (2).
The Cosmetic Ingredient Review expert group concluded in 2012 that this chemical compound did not present any significant toxicity and was therefore safe for use as a cosmetic ingredient (3).
References____________________________________________________
(1) Corazza M, Lauriola MM, Bianchi A, Zappaterra M, Virgili A. Irritant and sensitizing potential of eight surfactants commonly used in skin cleansers: an evaluation of 105 patients. Dermatitis. 2010 Sep-Oct;21(5):262-8.
(2) Mertens S, Gilissen L, Goossens A. Allergic contact dermatitis caused by cocamide diethanolamine. Contact Dermatitis. 2016 Jul;75(1):20-4. doi: 10.1111/cod.12580.
(3) Burnett CL, Bergfeld WF, Belsito DV, Hill RA, Klaassen CD, Liebler D, Marks JG Jr, Shank RC, Slaga TJ, Snyder PW, Andersen FA. Final report of the Cosmetic Ingredient Review Expert Panel on the safety assessment of cocamidopropyl betaine (CAPB). Int J Toxicol. 2012 Jul-Aug;31(4 Suppl):77S-111S. doi: 10.1177/1091581812447202.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Cocamidopropyl betaine Review Consensus 10 by Ark90 (12431 pt) | 2023-Jul-09 19:47 |
| Read the full Tiiip | (Send your comment) |
Cocamidopropyl betaine is a pseudo-amphoteric chemical compound, zwitterionic, with a quaternary ammonium cation that is industrially produced from coconut oil and dimethylaminopropylamine.
The name describes the structure of the molecule:
Cocamidopropyl is a compound derived from coconut oil and dimethylaminopropylamine. It contains a long chain of carbon atoms (from coconut oil fatty acids), a nitrogen atom (N) and three hydrogen atoms forming an amine group (-NH2).
betaine is a compound known as zwitterion, which has positive and negative charges but is neutral overall. In Cocamidopropyl betaine, the betaine part of the molecule is derived from the cocamidopropyl part by replacing one of the hydrogen atoms of the amine group with a carboxyl group (-COO-).
The synthesis process takes place in different steps:
It occurs as a clear to pale yellow transparent liquid or as a fine white powder. It dissolves easily in water (10 % solution), has an acid reaction with a pH of 5-7.

What it is used for and where it is used
Cosmetics
It is a surfactant (removes dirt particles) of synthetic origin and is used in cosmetics and body cleansers with an antimicrobial and foaming function. Softens hair and reduces static electricity in conditioners. Thickener in personal care products and detergents. Improves the conditioning functions of other surfactants, performs well against water hardness, is antistatic and biodegradable. Good compatibility with other amphoteric surfactants and cationic, anionic, non-ionic surfactants.
Cocamidopropyl betaine is one of the most common chemical compounds used in cosmetics and personal hygiene in detergents, liquid soaps, shampoos, eye make-up products, make-up removers, bath gels, contact lens solutions, roll-on deodorants. In shampoos the recommended dosage is 3-9%, while in cosmetics 1-2% is sufficient.
It has a significant number of INCI functions:
However, the presence of salt in solutions containing Cocamidopropyl betaine reduces their ability to lower surface tension, decreases the critical micelle concentration (mol/dm3) and increases absorption parameters (1).
Medical
In medicine and pharmaceuticals it is used in preparations for treating acne, exfoliating and peel-off products, anti-dandruff products etc..
Other uses
The objectives of this in vitro study were: a) to determine the effects of the waiting period of chlorhexidine (CHX) rinse after the use of fluoride toothpaste and b) to further determine the effect of the surfactant in the toothpaste [sodium dodecyl sulfate (SDS) or Cocamidopropyl betaine (CAPB)] on the remineralisation of the caries lesion associated with CHX rinse. The absence of CHX as an adjunct to fluoride toothpastes resulted in greater remineralisation of enamel lesions than the immediate use of CHX treatment for toothpastes with SDS and CAPB. CAPB toothpastes indicated significantly greater remineralisation than SDS toothpastes and may be recommended for patients at high risk of caries. A waiting time of 30 minutes for CHX treatment is recommended after brushing (2).
Safety
Cocamidopropyl betaine is normally among the least allergenic preservative chemical compounds, however its relative allergenicity appears to be attributed to its impurities dimethylaminopropylamine and cocamidopropyl dimethylamine and typically manifests as hand dermatitis (3).
Repeated and prolonged use of surfactants can cause irritation and allergic contact dermatitis. This study reports the frequency of positive patch test results for surfactants tested on a range of North American contact dermatitis screens, including cocamidopropyl betaine (CAPB), amidoamine (AA), dimethylaminopropylamine (DMAPA), oleamidopropyl dimethylamine (OPD) and cocamide diethanolamide (CDEA ) and correlations of positive reactions between CAPB and the other surfactants. This was a retrospective analysis of 10,877 patch patients tested between 2009 and 2014 for the surfactants CAPB, AA, DMAPA, OPD and CDEA. The frequencies of positive reactions to these surfactants and trends in reactivity among the surfactants analysed were calculated. OPD had the highest rate of positive reactions to the patch (2.3%) followed by DMAPA (1.7%) and CAPB (1.4%). AA and CDEA had the lowest rate of positive reactions (0.8%). There was a high degree of overlap in positive patch tests between the surfactants. CDEA was the least likely to co-react with another surfactant (4).
The degradation of the synthetic amphoteric surfactant, Cocamidopropyl betaine (CAPB) and its toxicity to the marine macroalgae, Ulva lactuca, were evaluated using different physiological test endpoints over different exposure periods of up to 120 hours .... The observations suggest that both CAPB and its metabolites are intrinsically toxic to U. lactuca. The results of this study are discussed in terms of the environmental consequences of the application of CAPB to the control of harmful blooms on algae (5).
Cocamidopropyl betaine (CAPB) and related amidopropylbainins are zwitterions mainly used as surfactants in cosmetics. These cosmetic ingredients are similar in their chemistry, particularly with regard to the presence of 3,3-dimethylamino-propylamine (DMAPA) and amidopropyl dimethylamine fatty acid (amidoamine) impurities, which are known sensitisers. The CIR Panel concluded that since these ingredients have no other significant toxicity, when formulated to be non-sensitising (i.e. can be based on a quantitative risk assessment), these ingredients are safe for use as cosmetic ingredients in the use and concentration practices of this safety assessment (6).
The most relevant studies on the subject have been selected with a summary of their contents:
Cocamidopropyl betaine studies
Typical optimal commercial product characteristics Cocamidopropyl betaine
| Appearance | Light yellow clear liquid |
| Melting point | < −10 °C (14 °F; 263 K) -50°C |
| Boiling point | 100°C 120°C |
| Flash point | 94°C |
| Solid Content | 35% ±1% 40% 45% |
| Free amine content | 0.5% max |
| Solid content | 35.0% min. |
| Active matter | 28.0% min. |
| pH value (5% aq.solution, 25℃) | 5-7 |
| Sodium chloride content | 7.0% max |
| PSA | 121.27000 |
| Free monochloroacetic acid | Max.100ppm |
| Sodium chloride | 6.0-7.0% Max |
| Free amine | 0.5% Max |
| Spec.gravity at 20℃ | 1.045-1.070 |
 |  |
 |  |
Synonyms :
References_______________________________________________________________________
(1) Staszak K, Wieczorek D, Michocka K. Effect of Sodium Chloride on the Surface and Wetting Properties of Aqueous Solutions of Cocamidopropyl Betaine. J Surfactants Deterg. 2015;18(2):321-328. doi: 10.1007/s11743-014-1644-8.
(2) Almohefer SA, Levon JA, Gregory RL, Eckert GJ, Lippert F. Caries lesion remineralization with fluoride toothpastes and chlorhexidine - effects of application timing and toothpaste surfactant. J Appl Oral Sci. 2018 Jun 11;26:e20170499. doi: 10.1590/1678-7757-2017-0499.
(3) Suuronen K, Pesonen M, Aalto-Korte K. Occupational contact allergy to cocamidopropyl betaine and its impurities. Contact Dermatitis. 2012 May;66(5):286-92. doi: 10.1111/j.1600-0536.2011.02036.x.
(4) Fowler JF Jr, Shaughnessy CN, Belsito DV, DeKoven JG, Deleo VA, Fransway AF, Maibach HI, Marks JG, Mathias CG, Pratt M, Sasseville D, Taylor JS, Warshaw EM, Zirwas MJ, Zug KA, Lorenz D. Cutaneous Delayed-Type Hypersensitivity to Surfactants. Dermatitis. 2015 Nov-Dec;26(6):268-70. doi: 10.1097/DER.0000000000000150.
(5) Vonlanthen S, Brown MT, Turner A. Toxicity of the amphoteric surfactant, cocamidopropyl betaine, to the marine macroalga, Ulva lactuca. Ecotoxicology. 2011 Jan;20(1):202-7. doi: 10.1007/s10646-010-0571-3.
(6) Burnett CL, Bergfeld WF, Belsito DV, Hill RA, Klaassen CD, Liebler D, Marks JG Jr, Shank RC, Slaga TJ, Snyder PW, Andersen FA. Final report of the Cosmetic Ingredient Review Expert Panel on the safety assessment of cocamidopropyl betaine (CAPB). Int J Toxicol. 2012 Jul-Aug;31(4 Suppl):77S-111S. doi: 10.1177/1091581812447202.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips about this object in __Italiano (2)
Component type: Chemical Main substances:
Last update: 2021-12-24 19:32:10 | Chemical Risk: |