App "Tiiips Ingredients Pro", Live Healthier!


App "Tiiips Ingredients Pro", Live Healthier!


| "Descrizione" about Hydrolyzed barley protein Review Consensus 9 by Nat45 (5698 pt) | 2023-Aug-06 17:38 |
| Read the full Tiiip | (Send your comment) |
Hydrolyzed Barley Protein refers to a product obtained by breaking down the proteins found in barley into smaller peptide fragments and amino acids.
The name defines the structure of the molecule
Description of raw materials used in production
Step-by-step industrial chemical synthesis process
What it is for and where
Cosmetics
Skin conditioning agent. It is the mainstay of topical skin treatment as it has the function of restoring, increasing or improving skin tolerance to external factors, including melanocyte tolerance. The most important function of the conditioning agent is to prevent skin dehydration, but the subject is rather complex and involves emollients and humectants that can be added in the formulation.
Hair conditioning agent. A significant number of ingredients with specific and targeted purposes may co-exist in hair shampoo formulations: cleansers, conditioners, thickeners, matting agents, sequestering agents, fragrances, preservatives, special additives. However, the indispensable ingredients are the cleansers and conditioners as they are necessary and sufficient for hair cleansing and manageability. The others act as commercial and non-essential auxiliaries such as: appearance, fragrance, colouring, etc. Hair conditioning agents have the task of increasing shine, manageability and volume, and reducing static electricity, especially after treatments such as colouring, ironing, waving, drying and brushing. They are, in practice, dispersants that may contain cationic surfactants, thickeners, emollients, polymers. The typology of hair conditioning agents includes: intensive conditioners, instant conditioners, thickening conditioners, drying conditioners. They can perform their task generally accompanied by other different ingredients.
Commercial applications
Skin products: Used in creams and lotions for their ability to moisturize and improve skin's elasticity.
Hair products: Found in shampoos, conditioners, and hair treatments, where they contribute to strengthening, moisturizing, and adding shine to the hair.
Properties
Moisturizing: Hydrolyzed barley proteins help retain moisture in the skin and hair, improving hydration.
Strengthening: They can increase hair's resistance to damage, making it stronger and less prone to breakage.
Elasticizing: They can improve the elasticity of the skin, making it firmer and more youthful.
Medical
Nutritional Supplements: Hydrolyzed Barley Protein is often used as a source of protein in nutritional supplements. Its constituent amino acids may be beneficial in certain medical scenarios, such as in patients with malnutrition or those who have difficulty digesting whole proteins.
Skin Care: Some studies have explored the application of hydrolyzed barley protein in skin care products. The peptides and amino acids in hydrolyzed barley protein may help moisturize and nourish the skin, promoting healing and providing anti-aging effects. There might be potential applications in the treatment of certain skin conditions, although more research is needed to confirm this.
Gastrointestinal Health: Barley itself is known for its high fiber content and potential gastrointestinal health benefits. The hydrolyzed form might be used in specific medical diets or as a therapeutic aid for individuals with digestive disorders.
Allergenicity Considerations: It is worth noting that hydrolyzed barley protein might not be suitable for individuals with barley or gluten allergies. Care must be taken when considering its use in medical applications for these populations.
Potential Development in Drug Delivery: There might be potential for hydrolyzed barley protein in the development of novel drug delivery systems, using the protein as a carrier for medication, although this is likely to be in early stages of research.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Hydrolyzed linseed seed by Nat45 (5698 pt) | 2023-Aug-05 17:21 |
| Read the full Tiiip | (Send your comment) |
Hydrolyzed Linseed Seed Is often used in cosmetics and personal care products. It is derived from the linseed.
The name defines the structure of the ingredient
Hydrolyzed. In the context of skincare and haircare products, hydrolyzed means the ingredient has been broken down into smaller units through a chemical reaction with water (a process known as hydrolysis). This process can make the ingredient more easily absorbed by the skin or hair.
Linseed Seed. Linseed is another name for flaxseed. Flaxseeds are a rich source of fatty acids and other nutrients, and they have been used in various forms in natural health and beauty products for their moisturizing and soothing properties.

Description of raw materials used in production
The raw materials for the production of hydrolyzed linseed are linseeds. These seeds are grown, harvested, and used in the hydrolysis process.
Extraction process
What it is for and where
Cosmetics
Skin conditioning agent. It is the mainstay of topical skin treatment as it has the function of restoring, increasing or improving skin tolerance to external factors, including melanocyte tolerance. The most important function of the conditioning agent is to prevent skin dehydration, but the subject is rather complex and involves emollients and humectants that can be added in the formulation.
Commercial applications
Skin products: Due to its moisturizing and soothing properties, it's used in a variety of skin products like creams, lotions, and serums.
Hair products: Hydrolyzed linseed seed is known for its hair benefits, making it a common ingredient in conditioners, hair masks, and styling products.
Anti-aging products: Due to its richness in antioxidants, it's used in anti-aging products to combat free radical damage to the skin.
Properties
Moisturizing: Helps retain moisture in the skin, helping to keep it hydrated.
Soothing: Can calm irritated skin, thanks to its anti-inflammatory properties.
Hair nourishing: Hydrolyzed linseed seed is rich in omega-3 fatty acids, which can nourish and fortify the hair.
As with all skincare and haircare products, it's essential to do a patch test before incorporating them into your routine to ensure you don't have any allergies or sensitivities.
Medical
In the seeds there are interesting antioxidant components such as the phenylpropanoid compounds, vanillic acid, vanillin, coumaric acid, ferulic acid and are the richest source of alpha-linolenic acid as well as an excellent source of dietary fibers.
The flax stem is the main source of cellulose-rich fibres used by the textile industry for the production of bed linen. Its seed oil (linseed) is beneficial for human health due to the presence of a high amount of omega-3 fatty acids. In addition, linseed oil is used in the preparation of many industrial solvents (1).
Flax contains about 34% oil and a high content of α-linolenic acid (> 50%) makes it a common feed ingredient for the enrichment of n-3 fatty acid (2).
It also contains mucilosis polysaccharides (neutral polysaccharides and acids composed mainly of galacturonic acid) (3).
References_________________________________________________________________________
(1) Shivaraj SM, Deshmukh RK, Rai R, Bélanger R, Agrawal PK, Dash PK Genome-wide identification, characterization, and expression profile of aquaporin gene family in flax (Linum usitatissimum). Sci Rep. 2017 Apr 27;7:46137. doi: 10.1038/srep46137.
(2) Cherian G, Quezada N. Egg quality, fatty acid composition and immunoglobulin Y content in eggs from laying hens fed full fat camelina or flax seed. J Anim Sci Biotechnol. 2016 Mar 3;7:15. doi: 10.1186/s40104-016-0075-y. eCollection 2016.
(3) European Scientific Cooperative on Phytotherapy. Lini semen. 2nd ed. New York: Thieme; 2003. ESCOP Monographs; pp. 290–6.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Camellia japonica flower extract by Nat45 (5698 pt) | 2023-Aug-05 16:26 |
| Read the full Tiiip | (Send your comment) |
Camellia japonica flower extract
The name describes the structure of the molecule
"Camellia japonica flower extract" is an extract derived from the flowers of the Camellia japonica plant, often known as Japanese camellia. It doesn't describe a single molecular structure, but rather a complex mix of various molecules, including flavonoids, phenolic acids, saponins, and other bioactive compounds.

Description of raw materials used in production
The primary raw material for producing Camellia japonica flower extract are the flowers. These flowers are cultivated, harvested, and used for the production of the extract.
Extraction process
What it is for and where
Cosmetics
Skin conditioning agent - Emollient. Emollients have the characteristic of enhancing the skin barrier through a source of exogenous lipids that adhere to the skin, improving barrier properties by filling gaps in intercorneocyte clusters to improve hydration while protecting against inflammation. In practice, they have the ability to create a barrier that prevents transepidermal water loss. Emollients are described as degreasing or refreshing additives that improve the lipid content of the upper layers of the skin by preventing degreasing and drying of the skin. The problem with emollients is that many have a strong lipophilic character and are identified as occlusive ingredients; they are oily and fatty materials that remain on the skin surface and reduce transepidermal water loss. In cosmetics, emollients and moisturisers are often considered synonymous with humectants and occlusives.
Skin conditioning agent. It is the mainstay of topical skin treatment as it has the function of restoring, increasing or improving skin tolerance to external factors, including melanocyte tolerance. The most important function of the conditioning agent is to prevent skin dehydration, but the subject is rather complex and involves emollients and humectants that can be added in the formulation.
CAS 223748-13-8
Commercial applications
Skin care products: Camellia japonica extract is used in a variety of skin care products, including creams, lotions, and serums, due to its moisturizing and protective properties.
Hair products: Given its ability to moisturize and nourish, it can be used in shampoos and conditioners.
Anti-aging products: Due to its richness in antioxidants, it's used in anti-aging products to combat free radical damage to the skin.
Properties
Moisturizing: Helps to keep the skin hydrated, making it soft and smooth.
Antioxidant: Contains antioxidants that protect the skin from free radical damage.
Protective: Can help protect the skin from environmental damage, such as pollution and UV rays.
Medical
Studies
Camellia species are considered in the scientific literature to be natural sources of antioxidant compounds and in particular with increased activity in flowers where a relevant presence of phenolic acids and flavonol glycosides has been noted (1).
As antibiotic resistance has become a worldwide problem, research tends to turn to natural sources, and many studies have evaluated the antimicrobial activity of Camellia japonica against Gram-negative and Gram-positive bacteria (2).
References_____________________________________________________________________
(1) Lee, H. H., Cho, J. Y., Moon, J. H., & Park, K. H. (2011). Isolation and identification of antioxidative phenolic acids and flavonoid glycosides from Camellia japonica flowers. Horticulture, Environment, and Biotechnology, 52, 270-277.
Abstract. The ethyl acetate (EtOAc) layer of the hot water extracts of Camellia japonica flowers was found to have higher 1,1-diphenyl-2-picrylhydrazyl (DPPH) radical-scavenging activity than the other solvent layers. Nine phenolic compounds were isolated and purified from the EtOAc layer by Sephadex LH-20 column chromatography and octadecyl silane-high performance liquid chromatography using a guided DPPH radical-scavenging assay. The isolated compounds were identified as 3,4,5-trihydroxybenzoic acid (1), 3,4-dihydroxybenzoic acid (2), 4-hydroxybenzoic acid (3), 2,3-digalloyl-O-α-D-glucopyranoside (4), 2,3-digalloyl-O-β-D-glucopyranoside (5), quercetin 3-O-β-D-galactopyranoside (6), quercetin 3-O-β-D-glucopyranoside (7), kaempferol 3-O-β-D-galactopyranoside (8), and kaempferol 3-O-β-D-glucopyranoside (9), based on mass spectrometry and nuclear magnetic resonance. Four compounds (6–9) had been previously identified in the leaves of this plant, but other compounds (1–5) were newly isolated from this plant. Their DPPH radical-scavenging activities based on the 50% scavenging concentration decreased in the following order: 4 = 5 (4.7 μM) > 1 (9.8 μM) > 6 = 7 (8.2 μM) > α-tocopherol (24.7 M) > ascorbic acid (25.1 μM) > 2 (35.6 M) > 3 = 8 = 9 (> 250 μM). Quercetin glycosides (6, 7), gallic acid (1) and its glucosides (4, 5) showed higher DPPH radical-scavenging activities than other compounds. These results indicate that the antioxidant effect of C. japonica flowers may be attributable to quercetin glycosides and gallic acid derivatives. These isolated compounds will be useful in basic studies of plant physiology, food manufacturing, and biological function of C. japonica flowers.
(2) Kim KY, Davidson PM, Chung HJ. Antibacterial activity in extracts of Camellia japonica L. petals and its application to a model food system. J Food Prot. 2001 Aug;64(8):1255-60. doi: 10.4315/0362-028x-64.8.1255.
Abstract. The potential presence of naturally occurring antimicrobials in petals of Camellia japonica L., a member of the tea family, was investigated against foodborne pathogens in microbiological media and food. Petals of the camellia flower (C. japonica L.) were extracted with methanol and fractionated into basic, acidic, and neutral fractions. The acidic fraction (equivalent to 1.0 g of raw sample per disk) produced an inhibitory zone of 14 to 19 mm (diameter) in a disk assay against the pathogens Salmonella Typhimurium DT104, Escherichia coli O157:H7, Listeria monocytogenes, and Staphylococcus aureus on agar plates. Silica gel adsorption column chromatography, Sephadex LH-20 column chromatography, and preparative purification by high-pressure liquid chromatography were used to purify compounds in the fraction. The mass spectrum of the antibacterial compound isolated had a molecular ion (M+) of m/z 116 and showed good conformity with the spectrum of fumaric acid (HOOC-CH=CH-COOH). An aqueous extract from the petals of C. japonica L. had an inhibitory effect on growth of all pathogens at 37 degrees C in microbiological media by increasing the lag phase. None of the microorganisms was inhibited completely. Milk was used as a model food system. Aqueous extract at a concentration of 100 mg/ml was bacteriostatic against all the foodborne pathogens in the milk stored at 25 degrees C for up to 4 days.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Quinine monohydrochloride dihydrate Review Consensus 10 by Nat45 (5698 pt) | 2023-Jun-27 20:19 |
| Read the full Tiiip | (Send your comment) |
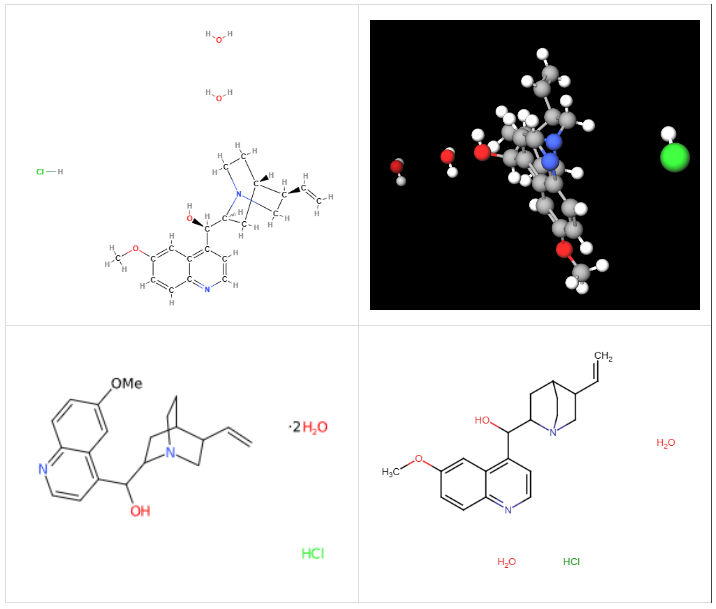
Quinine monohydrochloride dihydrate or Quinine monochloride dihydrate is a salt obtained chemically from quinine. Quinine is an alkaloid from the bark of the China tree, family Rubiaceae. Three salts are obtained from quinine: FL 14,011, FL 14,152 and FL 14,155, and the latter is what is called Quinine monochloride dihydrate. The specific properties of Quinine monochloride dihydrate, such as solubility and stability, may vary depending on storage and usage conditions.
Quinine occurs naturally in the bark of the cinchona tree and was the first effective western treatment for malaria.
The name defines the structure of the molecule:
The synthesis process takes place in several stages:
Monochlorinated quinine dihydrate is a solid at standard temperature and pressure (STP), with a melting point of 115.5 ºC. It is typically found in powder form. The color may vary depending on its purity and the specific production process, but it is typically white or off-white.

What it is used for and where
Food
It is used in drinks and some foods to impart a bitter taste.
Contraindications - studies
Glucose-6-phosphate dehydrogenase (G6PD) deficiency is the most common human enzyme defect that often appears in neonatal jaundice and/or haemolytic anaemia. G6PD haemolytic events are linked to exposure to a pro-oxidant agent. Here we report three cases of initial G6PD crises in breastfed infants with maternal consumption of a tonic drink containing quinine. Quinine was found in the breast milk of one of the mothers after consuming tonic water. The amount of quinine that is transmitted through breast milk appears to be sufficient to induce G6PD seizures in breastfed infants. Therefore, it is recommended to avoid consumption of quinine-containing drinks during breastfeeding in populations with a high prevalence of G6PD deficiency.
What is known:
What's new:
Quinine is a common cause of drug-induced thrombocytopenia and the most common cause of drug-induced thrombotic microangiopathy. Other quinine-induced systemic disorders have been described. In order to understand the full clinical spectrum of adverse reactions to quinine, the author of this study consulted 11 databases that provided sufficient data to allow evaluation of levels of evidence supporting a causal association with quinine. Three reviewers independently determined levels of evidence, including both immune-mediated and toxic adverse reactions. The main focus of this review was on acute, immune-mediated reactions. The source of quinine exposure, organ systems involved, severity of adverse reactions and patient outcomes were documented. One hundred and fourteen articles described 142 patients with definite or probable evidence of a causal association of quinine with acute, immune-mediated reactions. One hundred and nine (72%) reactions were caused by quinine pills; 28 (20%) by quinine-containing beverages; 12 (8%) by five other types of exposure.Quinine, even with minimal exposure from common beverages, can cause severe adverse reactions involving multiple organ systems. In patients with acute, multisystem disorders of unknown origin, an adverse reaction to quinine must be considered (2).
European legislation has dealt with this substance (3) in order to assess its effects on human health and, in the opinion of the Scientific Committee on Food (SCF) of 19 February 1988, no objections were raised, from a toxicological point of view, to the continued use of quinine in bitter drinks at current levels (up to a maximum of 100 mg/l). While not contesting this assessment, the Authority recommends a review of the toxicological database on quinine (4). Pending the re-evaluation of quinine, the use of three quinine salts (FL 14.011, FL 14.152 and FL 14.155) should be restricted to non-alcoholic and alcoholic beverages.
Synonyms :
References_____________________________________________________________________
(1) Bichali S, Brault D, Masserot C, Boscher C, Couec ML, Deslandes G, Pissard S, Leverger G, Vauzelle C, Elefant E, Rozé JC, Cortey A, Chenouard A. Maternal consumption of quinine-containing sodas may induce G6PD crises in breastfed children. Eur J Pediatr. 2017 Oct;176(10):1415-1418. doi: 10.1007/s00431-017-2998-5.
(2) Liles NW, Page EE, Liles AL, Vesely SK, Raskob GE, George JN. Diversity and severity of adverse reactions to quinine: A systematic review. Am J Hematol. 2016 May;91(5):461-6. doi: 10.1002/ajh.24314.
(3) «Report of the Scientific Committee for Food on Quinine.» (19 February 1988). In Food — Science and techniques. Reports of the Scientific Committee for Food (Twenty-first series).
(4) «Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food on a request from Commission on Flavouring Group Evaluation 35, (FGE.35) Three quinine salts from the Priority list from chemical group 30.»EFSA Journal (2008) 739, pagg. 1-18.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Starch aluminium octenyl succinate Review Consensus 9 by Nat45 (5698 pt) | 2023-Oct-22 17:37 |
| Read the full Tiiip | (Send your comment) |
Starch aluminium octenyl succinate (Aluminum Starch Octenylsuccinate) is obtained by the reaction of octenylsuccinic anhydride with starch and is the aluminium salt of the product of this reaction.
The name describes the structure of the molecule
Description of raw materials and their functions:
Starch is the main raw material and is used as a source of carbohydrates for polymer formation.
Aluminum may be added as a compound or catalyst to facilitate the chemical reaction.
Succinic acid is used as a reagent to form the succinate ester in starch.
Step-by-step summary of the industrial chemical synthesis process:
The compound "Starch Aluminium Octenyl Succinate" is commonly used in the food and pharmaceutical industries as a coating agent, binding agent, or stabilizer to enhance consistency, stability, and solubility characteristics of the final products.
It appears as a white powder or white granules.

What it is used for and where
Cosmetics
Absorbent. Absorbs substances dispersed or dissolved in aqueous solutions, water/oil, oil/water.
Anticaking agent. This chemical compound facilitates free flow and prevents aggregation or clumping of substances in a formulation by reducing the tendency of certain particles to stick together.
Viscosity control agent. It controls and adapts viscosity to the required level for optimal chemical and physical stability of the product and dosage in gels, suspensions, emulsions, solutions.
Normalmente viene inserito nelle creme in una percentuale che non supera il 30%.
Food
It is also used in the food industry as a modified food starch and is subject to certain restrictions with regard to heavy metal residues.
In the list of European food additives, aluminium starch octenyl succinate has the number E1452 as a thickener.
Safety
The EFSA Panel on Food Additives and Nutrient Sources Added to Food considers that there is no safety concern for the use of modified starches as food additives at the uses and use levels declared for the general population and that a numerical ADI is not necessary (1).
A warning should be given about the risk of aluminum intake, which cannot be ruled out as this ingredient can be found both in cosmetic products and in widely consumed foodstuffs such as bread, various baked goods (2)
In the minute quantities in which it is included in food or cosmetics, it is not a cause for concern. However, aluminium is a possible endocrine disruptor, so monitor any cumulative intake.
References________________________________________________________________________
(1) EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), Mortensen, A., Aguilar, F., Crebelli, R., Di Domenico, A., Dusemund, B., Frutos, M.J., Galtier, P., Gott, D., Gundert‐Remy, U. and Lambré, C., 2017. Re‐evaluation of oxidised starch (E 1404), monostarch phosphate (E 1410), distarch phosphate (E 1412), phosphated distarch phosphate (E 1413), acetylated distarch phosphate (E 1414), acetylated starch (E 1420), acetylated distarch adipate (E 1422), hydroxypropyl starch (E 1440), hydroxypropyl distarch phosphate (E 1442), starch sodium octenyl succinate (E 1450), acetylated oxidised starch (E 1451) and starch aluminium octenyl succinate (E 1452) as food additives. EFSA Journal, 15(10), p.e04911.
Abstract. Following a request from the European Commission, the EFSA Panel on Food Additives and Nutrient sources added to Food (ANS) was asked to deliver a scientific opinion on the re-evaluation of 12 modified starches (E 1404, E 1410, E 1412, E 1413, E 1414, E 1420, E 1422, E 1440, E 1442, E 1450, E 1451 and E 1452) authorised as food additives in the EU in accordance with Regulation (EC) No 1333/2008 and previously evaluated by JECFA and the SCF. Both committees allocated an acceptable daily intake (ADI) ‘not specified’. In humans, modified starches are not absorbed intact but significantly hydrolysed by intestinal enzymes and then fermented by the intestinal microbiota. Using the read-across approach, the Panel considered that adequate data on short- and long-term toxicity and carcinogenicity, and reproductive toxicity are available. Based on in silico analyses, modified starches are considered not to be of genotoxic concern. No treatment-related effects relevant for human risk assessment were observed in rats fed very high levels of modified starches (up to 31,000 mg/kg body weight (bw) per day). Modified starches (e.g. E 1450) were well tolerated in humans up to a single dose of 25,000 mg/person. Following the conceptual framework for the risk assessment of certain food additives, the Panel concluded that there is no safety concern for the use of modified starches as food additives at the reported uses and use levels for the general population and that there is no need for a numerical ADI. The combined exposure to E 1404–E 1451 at the 95th percentile of the refined (brand-loyal) exposure assessment scenario for the general population was up to 3,053 mg/kg bw per day. Exposure to E 1452 for food supplement consumers only at the 95th percentile was up to 22.1 mg/kg bw per day.
Safety assessment of starch-based personal care products: Nanocapsules and pickering emulsions. Marto J, Pinto P, Fitas M, Gonçalves LM, Almeida AJ, Ribeiro HM. Toxicol Appl Pharmacol. 2018 Mar 1;342:14-21. doi: 10.1016/j.taap.2018.01.018.
Final report on the safety assessment of aluminum starch octenylsuccinate. Nair B, Yamarik TA; Cosmetic Ingredient Review Expert panel. Int J Toxicol. 2002;21 Suppl 1:1-7. Review.
(2) EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS); Younes M, Aggett P, Aguilar F, Crebelli R, Dusemund B, Filipič M, Frutos MJ, Galtier P, Gott D, Gundert-Remy U, Kuhnle GG, Lambré C, Leblanc JC, Lillegaard IT, Moldeus P, Mortensen A, Oskarsson A, Stankovic I, Waalkens-Berendsen I, Wright M, Di Domenico A, van Loveren H, Giarola A, Horvath Z, Lodi F, Tard A, Woutersen RA. Re-evaluation of aluminium sulphates (E 520-523) and sodium aluminium phosphate (E 541) as food additives. EFSA J. 2018 Jul 27;16(7):e05372. doi: 10.2903/j.efsa.2018.5372.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Avobenzone Review Consensus 10 by Nat45 (5698 pt) | 2023-Jul-28 15:49 |
| Read the full Tiiip | (Send your comment) |
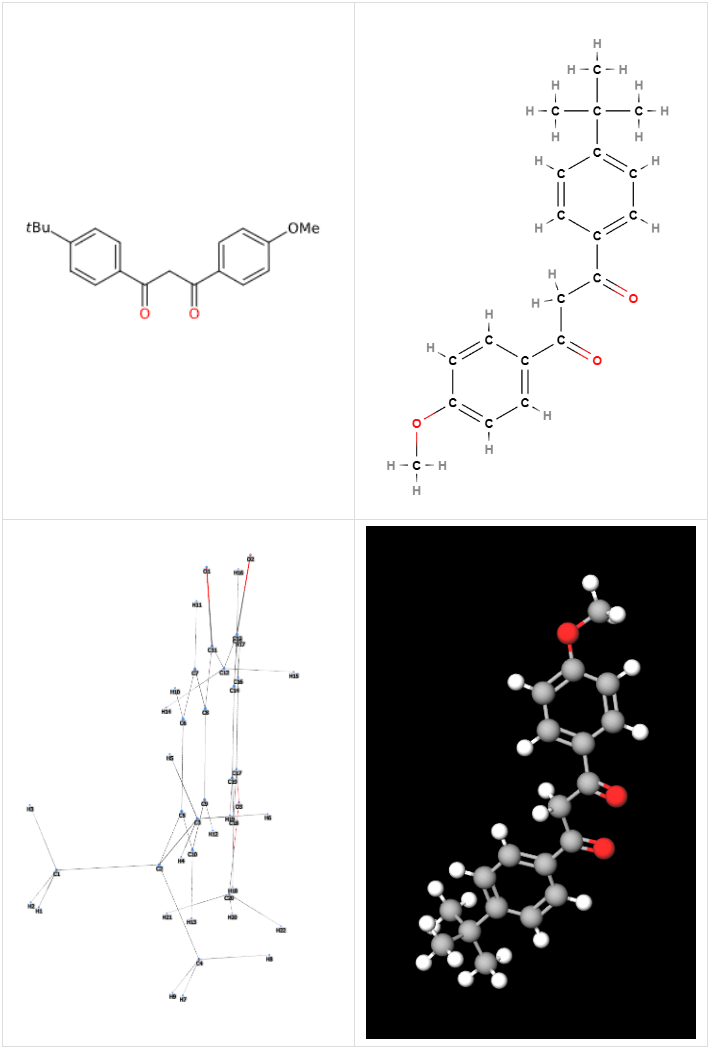
Avobenzone or Butyl Methoxydibenzoylmethane is an organic compound produced by the esterification of salicylic acid with butyl alcohol in the presence of an acid catalyst to produce butyl salicylate which, in a Claisen condensation reaction with benzoyl chloride or its derivatives, forms butyl methoxydibenzoylmethane. Subsequently, this product is purified using various techniques such as recrystallisation, solvent extraction and filtration to obtain the purest possible form of butyl methoxydibenzoylmethane.
The name describes the structure of the molecule
Description of raw materials used in production
Synthesis process
Butyl Methoxydibenzoylmethane and Avobenzone are the same chemical compound.
It appears as a white powder that is not very soluble in water, but soluble in organic solvents such as isopropyl alcohol, ethanol and oils.

What it is used for and where
Cosmetics
It is a restricted ingredient VI/8 as a Relevant Item in the Annexes of the European Cosmetics Regulation 1223/2009.
Butyl Methoxydibenzoylmethane is generally used in cosmetics as a UV filter, maximum absorption length of 360nm (1) and as an INCI ingredient has the following functions:
UV filter. It is the defining ingredient in sun creams that can mitigate the sun's ultraviolet (UV) radiation, which is a high risk factor for the development of skin cancer, erythema and photo-ageing.
UV absorber. It acts by intercepting ultraviolet light before it can cause damage by reducing its energy through dissipation and returning it to a lower energy state.
Light stabilizer. It prevents light from degrading light-sensitive components and slows down degradation reactions that have already begun. The mechanism is, in a way, similar to antioxidants and the effectiveness depends on the.complexity of the formulation and the density of the product.
The most relevant studies on this chemical compound have been selected with a summary of their contents:
Synonyms:
References____________________________________________
(1) Li X, Hu J, Chen L, Zhang W. The adsorption behaviour and photoprotection effect of UV filter absorbed on the surface of human hair. Int J Cosmet Sci. 2016 Feb;38(1):52-9. doi: 10.1111/ics.12247.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about Butyl Methoxydibenzoylmethane Review Consensus 10 by Nat45 (5698 pt) | 2023-Jul-28 15:50 |
| Read the full Tiiip | (Send your comment) |
Butyl Methoxydibenzoylmethane or Avobenzone is an organic compound produced by the esterification of salicylic acid with butyl alcohol in the presence of an acid catalyst to produce butyl salicylate which, in a Claisen condensation reaction with benzoyl chloride or its derivatives, forms butyl methoxydibenzoylmethane. Subsequently, this product is purified using various techniques such as recrystallisation, solvent extraction and filtration to obtain the purest possible form of butyl methoxydibenzoylmethane.
The name describes the structure of the molecule
Description of raw materials used in production
Synthesis process
Butyl Methoxydibenzoylmethane and Avobenzone are the same chemical compound.
It appears as a white powder that is not very soluble in water, but soluble in organic solvents such as isopropyl alcohol, ethanol and oils.

What it is used for and where
Cosmetics
It is a restricted ingredient VI/8 as a Relevant Item in the Annexes of the European Cosmetics Regulation 1223/2009.
Butyl Methoxydibenzoylmethane is generally used in cosmetics as a UV filter, maximum absorption length of 360nm (1) and as an INCI ingredient has the following functions:
UV filter. It is the defining ingredient in sun creams that can mitigate the sun's ultraviolet (UV) radiation, which is a high risk factor for the development of skin cancer, erythema and photo-ageing.
UV absorber. It acts by intercepting ultraviolet light before it can cause damage by reducing its energy through dissipation and returning it to a lower energy state.
Light stabilizer. It prevents light from degrading light-sensitive components and slows down degradation reactions that have already begun. The mechanism is, in a way, similar to antioxidants and the effectiveness depends on the.complexity of the formulation and the density of the product.
The most relevant studies on this chemical compound have been selected with a summary of their contents:

Synonyms:
References____________________________________________
(1) The adsorption behaviour and photoprotection effect of UV filter absorbed on the surface of human hair.
Li X, Hu J, Chen L, Zhang W.
Int J Cosmet Sci. 2016 Feb;38(1):52-9. doi: 10.1111/ics.12247.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips in __Italiano (26)
