App "Tiiips Ingredients Pro", Live Healthier!


App "Tiiips Ingredients Pro", Live Healthier!


| "Descrizione" about L-carnitine Review Consensus 10 by FRanier (9976 pt) | 2023-Jul-11 16:10 |
| Read the full Tiiip | (Send your comment) |
L-carnitine, chemically known as 3-hydroxy-4-(trimethylammonium)butanoate, is a quaternary ammonium compound involved in the metabolism of most mammals, plants and some bacteria.
The name defines the structure of the molecule:
In the human body, L-carnitine is synthesised from the amino acids lysine and methionine.
The synthesis process takes place in different steps:
It appears in the form of a white powder.

What it is used for and where
It is often used as a dietary supplement for its potential benefits in weight loss, brain function, heart health and exercise performance.
Medical
Carnitine is synthesised in many eukaryotic organisms and its biosynthesis is initiated by the methylation of lysine. It plays a key role in the metabolism of fatty acids in the mitochondria (1).
Carnitine, however, is not considered an essential nutrient and is present in many foods, mainly those from animal sources. Lysine and methionine are necessary ingredients for carnitine biosynthesis.
All body tissues can produce deoxy-carnitine but, in humans, the enzyme that enables the hydroxylation of deoxy-carnitine to carnitine is only found in the liver, brain and kidneys (2).
Cosmetics
It performs a number of interesting acts.
Anti-static agent. Static electricity build-up has a direct influence on products and causes electrostatic adsorption. The antistatic ingredient reduces static build-up and surface resistivity on the surface of the skin and hair.
Hair conditioning agent. A significant number of ingredients with specific and targeted purposes can co-exist in a hair shampoo: cleansers, conditioners, thickeners, mattifying agents, sequestering agents, fragrances, preservatives, special additives. However, the indispensable ingredients are the cleansers and conditioners as they are necessary and sufficient for hair cleansing and manageability. The others have commercial and non-essential accessory acts such as: appearance, perfume, colouring, etc. Hair conditioning agents have the task of increasing shine, manageability and volume, and reducing static electricity, especially after treatments such as colouring, ironing, waving, drying and brushing. They are, in practice, dispersants that may contain cationic surfactants, thickeners, emollients, polymers. The typology of hair conditioning agents includes: intensive conditioners, instant conditioners, thickening conditioners, drying conditioners.
Surfactant - Cleansing agent. Cosmetic products used to cleanse the skin utilise the surfactant action that produces a lowering of the surface tension of the stratum corneum, facilitating the removal of dirt and impurities.
Surfactant - Foam booster. This has the function of introducing gas bubbles into the water for a purely aesthetic factor, which does not affect the cleansing process, but only satisfies the commercial aspect of the cleanser by helping to spread the cleanser on the hair. This helps in the commercial success of a shampoo formulation. Since sebum has an inhibiting action on the bubble, more foam is produced in the event of a second shampoo.
Viscosity-increasing agent, aqueous. Since viscosity is important to increase the chemical and physical stability of the product, Viscosity Enhancing Agent, aqueous is an important dosage factor in gels, suspensions, emulsions, solutions. Increasing viscosity makes formulations less sedimentary and more homogeneously thickened.
The most relevant studies on carnitine have been selected with a summary of their contents:

References____________________________________________________________________
(1) Bremer J. Carnitine—metabolism and functions. Physiol. Rev. 1983;63:1420–80.
(2) Jacob C, Belleville F. L-carnitine: metabolism, functions and value in pathology. Pathol Biol (Paris). 1992 Nov;40(9):910-9.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about E433 Review Consensus 9 by FRanier (9976 pt) | 2023-Jul-05 19:19 |
| Read the full Tiiip | (Send your comment) |
E433 (Polysorbate 80 or Polyoxyethylene sorbitan monooleate) is a non-toxic non-ionic detergent widely used for solubilizing and emulsifying hydrophobic pharmaceuticals, cosmetics, and food additives (1).
The name describes the structure of the molecule:
The synthesis process takes place in several stages.
What it is used for and where
Cosmetics
It is used as a surfactant, stabilizer and emulsifier in the composition of cosmetics and has these INCI functions:
Denaturant. The ionic or polar molecules of this ingredient included in formulations that interact with protein groups, modulate the properties of the solution to suit specific needs.
Surfactant - Cleansing agent. Cosmetic products used to cleanse the skin utilise the surface-active action that produces a lowering of the surface tension of the stratum corneum, facilitating the removal of dirt and impurities.
Surfactant - Emulsifying agent. Emulsions are thermodynamically unstable and are used to soothe or soften the skin and emulsify, so they need a specific, stabilising ingredient. This ingredient forms a film, lowers the surface tension and makes two immiscible liquids miscible. A very important factor affecting the stability of the emulsion is the amount of the emulsifying agent. Emulsifiers have the property of reducing the oil/water or water/oil interfacial tension, improving the stability of the emulsion and also directly influencing the stability, sensory properties and surface tension of sunscreens by modulating the filmometric performance.
Food
Ingredient included in the list of European food additives such as E433 with emulsifier function and with name
Labeled as E433 in the list of food additives.
How it is composed
The fatty acid (FA) composition of polysorbate 80 (PS80), a sorbitan oleic acid ester copolymerized with about 20mole of ethylene oxide, is typically characterized by gas chromatography. Here, an alternative method was developed. After saponification with potassium hydroxide the FA fraction was collected with liquid-liquid extraction using methyl-tert-butyl ether. HPLC in combination with a Corona® charged aerosol detector (CAD) was applied for the separation and detection. The method was fully validated in terms of specificity, repeatability, limits of quantification, linearity, range, accuracy and robustness. The characterization of 16 different PS80 batches demonstrated variability regarding their FA composition, with e.g. the amount of oleic acid ranging from 67.8±0.7% to 96.6±1.4%. Furthermore, this study identified petroselinic acid, a double-bond positional isomer to oleic acid in all batches, an FA not known to pharmacopoeias at present. In addition, 11-hydroxy-9-octadecenoic acid, an oxidation product of oleic acid was identified. Structure elucidation was performed by means of HPLC-MS/MS. In addition, the method was expanded to the evaluation of the free FAs. Having determined the entire FA composition, the acid value according to EP and USP can be calculated (2)
Medical - Studies


Safety
Polysorbate 80 has been involved in isolated cases of allergy in the form of contact dermatitis caused by topical drugs and in other cases after parenteral administration causing generalized reactions such as urticaria-angioedema and anaphylaxis (7).
The Joint Food and Agriculture Organization of the United Nations (FAO)/World Health Organization (WHO)Expert Committee on Food Additives (JECFA)derived an Acceptable Daily Intake (ADI) of 25mg/kg body weight (bw)/day (group ADI for polysorbates 20, 40, 60, 65 and 80) and the Scientific Committee on Food (SCF) derived a group ADI of 10mg/kg bw/day. Small amounts of polyoxyethylene sorbitansare absorbed. Similar toxicokinetics would be expected for all polysorbates based on their similarities in structure and metabolic fate. The acute toxicity is very low. There is no concern regarding genotoxicity, carcinogenicity or developmental toxicity. From a limited number of studies,there is no indication of reproductive toxicity. The Panel recommended that the maximum limits for the impurities of toxic elements (arsenic, lead, cadmium and mercury) in the EC specification for polysorbates (E432–E436) should be revised in order to ascertain that polysorbates (E432–E436) as food additives will not be a significant source of exposure to those toxic elements in food (8).

Synonyms :
Tween(R) 80, Polyoxyethylene 20 sorbitan monooleate, 2-hydroxyethyl 2-deoxy-3,5-bis-O-(2-hydroxyethyl)-6-O-{2-[(9E)-octadec-9-enoyloxy]ethyl}hexofuranoside, sorbate80, Polyoxyethylene 20 sorbitan monooleate, EG Coolant, Sorbitan, mono-9-octadecenoate, poly(oxy-1,2-ethanediyl) derivs, 2-hydroxyethyl 2-deoxy-3,5-bis-O-(2-hydroxyethyl)-6-O-[2-(oleoyloxy)ethyl]hexofuranoside, 2-[2-[3,5-bis(2-hydroxyethoxy)oxolan-2-yl]-2-(2-hydroxyethoxy)ethoxy]ethyl
References______________________________________________________
(1) Narang AS, Delmarre D, Gao D. Stable drug encapsulation in micelles and microemulsions. Int J Pharm. 2007 Dec 10;345(1-2):9-25. doi: 10.1016/j.ijpharm.2007.08.057.
Abstract. Oral absorption of hydrophobic drugs can be significantly improved using lipid-based non-particulate drug delivery systems, which avoid the dissolution step. Micellar and microemulsion systems, being the most dispersed of all, appear the most promising. While these systems show high drug entrapment and release under sink conditions, the improvement in oral drug bioavailability is often unpredictable. The formulation and drug-related biopharmaceutical aspects of these systems that govern oral absorption have been widely studied. Among these, preventing drug precipitation upon aqueous dilution could play a predominant role in many cases. Predictive ability and quick methods for assessment of such problems could be very useful to the formulators in selecting lead formulations. This review will attempt to summarize the research work that could be useful in developing these tools.
Williams, H.D.; Trevaskis, N.L.; Charman, S.A.; Shanker, R.M.; Charman, W.N.; Pouton, C.W.; Porter, C.J.H Strategies to address low drug solubility in discovery and development. . Pharmacol. Rev. 2013, 65, 315–499
(2) Ilko D, Braun A, Germershaus O, Meinel L, Holzgrabe U. Fatty acid composition analysis in polysorbate 80 with high performance liquid chromatography coupled to charged aerosol detection. Eur J Pharm Biopharm. 2015 Aug;94:569-74. doi: 10.1016/j.ejpb.2014.11.018.
(3) Navari RM. HTX-019: polysorbate 80- and synthetic surfactant-free neurokinin 1 receptor antagonist for chemotherapy-induced nausea and vomiting prophylaxis. Future Oncol. 2019 Jan;15(3):241-255. doi: 10.2217/fon-2018-0577.
(4) Li Y, Wu M, Zhang N, Tang C, Jiang P, Liu X, Yan F, Zheng H. Mechanisms of enhanced antiglioma efficacy of polysorbate 80-modified paclitaxel-loaded PLGA nanoparticles by focused ultrasound. J Cell Mol Med. 2018 Sep;22(9):4171-4182. doi: 10.1111/jcmm.13695.
(5) Sloup RE, Cieza RJ, Needle DB, Abramovitch RB, Torres AG, Waters CM. Polysorbates prevent biofilm formation and pathogenesis of Escherichia coli O104:H4. Biofouling. 2016 Oct;32(9):1131-1140. doi: 10.1080/08927014.2016.1230849.
(6) Malinowski AM, McClarty BM, Robinson C, Spear W, Sanchez M, Sparkes TC, Brooke JS. Polysorbate 80 and polymyxin B inhibit Stenotrophomonas maltophilia biofilm. Diagn Microbiol Infect Dis. 2017 Feb;87(2):154-156. doi: 10.1016/j.diagmicrobio.2016.11.008.
(7) Palacios Castaño MI, Venturini Díaz M, Lobera Labairu T, González Mahave I, Del Pozo Gil MD, Blasco Sarramián A. Anaphylaxis Due to the Excipient Polysorbate 80. J Investig Allergol Clin Immunol. 2016;26(6):394-396. doi: 10.18176/jiaci.0109.
(8) EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS), 2015. Scientific Opinion on the re‐evaluation of polyoxyethylene sorbitan monolaurate (E 432), polyoxyethylene sorbitan monooleate (E 433), polyoxyethylene sorbitan monopalmitate (E 434), polyoxyethylene sorbitan monostearate (E 435) and polyoxyethylene sorbitan tristearate (E 436) as food additives. Efsa Journal, 13(7), p.4152.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about E1105 Review Consensus 9 by FRanier (9976 pt) | 2023-Apr-28 09:11 |
| Read the full Tiiip | (Send your comment) |
Lysozyme is a lytic enzyme-type protein found in the human body (liver, spleen. saliva) eggs and other animal tissues, a peptide with antimicrobial activity.
Industrially it appears in the form of a white powder.

What it is used for and where
Food
Ingredient included in the list of European food additives as E1105 with a preservative function and is added to cheeses (e.g. grana padano) to maintain their integrity and to counteract Clostridia spp. bacteria.
Medical
It acts as an antioxidant, anti-inflammatory and antibacterial agent especially against Gram-positive and Gram-negative bacteria (1), acts as a protective agent in Alzheimer's disease (2), in the treatment of infections by Staffilococcus Aureus bacteria (3) and is an effective substitute for chlorhexidine and benzidine in the treatment of acute pharyngitis (4).
Cosmetics
Skin conditioning agent. It is the mainstay of topical skin treatment as it has the function of restoring, increasing or improving skin tolerance to external factors, including melanocyte tolerance. The most important function of the conditioning agent is to prevent skin dehydration, but the subject is rather complex and involves emollients and humectants that can be added in the formulation.
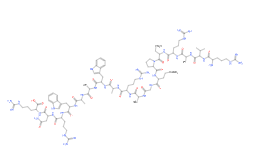 | 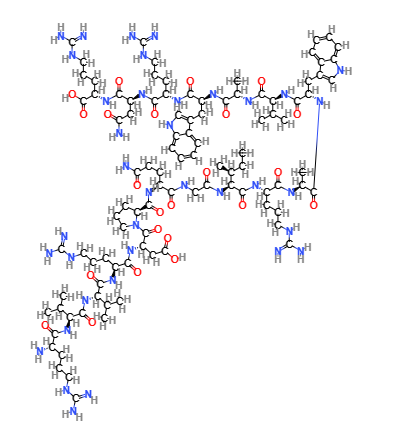 |
 | 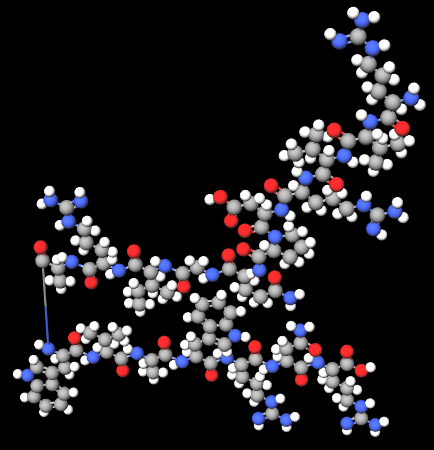 |
References_________________________________________________________________________
(1) Vilcacundo R, Méndez P, Reyes W, Romero H, Pinto A, Carrillo W. Antibacterial Activity of Hen Egg White Lysozyme Denatured by Thermal and Chemical Treatments. Sci Pharm. 2018 Oct 30;86(4):E48. doi: 10.3390/scipharm86040048.
Abstract. The aim of this study was to increase the antibacterial spectrum of modified hen egg white lysozyme (HEWL) with thermal and chemical treatments against Gram-negative bacteria. The antibacterial activity of heat-denatured HEWL and chemical denatured HEWL against Gram-negative and Gram-positive bacteria was evaluated in 15 h of incubation tests. HEWL was denatured by heating at pH 6.0 and pH 7.0 and chemical denaturing was carried out for 1.0, 1.5, 2.0, and 4.0 h with DL-Dithiothreitol (DTT). HEWL modified by thermal and chemical treatments was characterized using the sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis method. Heat-denatured HEWL lytic activity against Micrococcus lysodeikticus lessened with increasing temperature and time of incubation with the chemical agent (DTT). The loss of lytic activity in modified HEWL suggests that the mechanism of action of the antibacterial activity is not dependent on the lytic activity. Thermal and chemical treatments of HEWL enabled the production of oligoforms and increased antibacterial activity over a wider spectrum. Heat-denatured HEWL at pH 6.0 and chemically-denatured HEWL increased the HEWL antibacterial spectrum against Gram-negative bacteria (Escherichia coli ATCC 25922). HEWL at 120 °C and pH 6.0 (1.0 mg/mL) inhibited 78.20% of the growth of E. coli. HEWL/DTT treatment for 4.0 h (1.0 mg/mL) inhibited 68.75% of the growth E. coli. Heat-denatured HEWL at pH 6.0 and pH 7.0 and chemically-denatured HEWL (1.0, 1.5, 2.0, and 4.0 h with DTT) were active against Gram-positive bacteria (Staphylococcus carnosus CECT 4491T). Heat-denatured and chemical-denatured HEWL caused the death of the bacteria with the destruction of the cell wall. LIVE/DEAD assays of fluorescent dye stain of the membrane cell showed membrane perturbation of bacteria after incubation with modified HEWL. The cell wall destruction was viewed using electron microscopy. The results obtained in this study suggest that heat-denatured HEWL at pH 6.0 and chemical-denatured HEWL treatments increase the HEWL antibacterial activity against Gram-negative bacteria.
(2) Helmfors L, Boman A, Civitelli L, Nath S, Sandin L, Janefjord C, McCann H, Zetterberg H, Blennow K, Halliday G, Brorsson AC, Kågedal K. Protective properties of lysozyme on β-amyloid pathology: implications for Alzheimer disease. Neurobiol Dis. 2015 Nov;83:122-33. doi: 10.1016/j.nbd.2015.08.024.
(3) Chen, L.L., Shi, W.P., Zhang, T.D., Zhou, Y.Q., Zhao, F.Z., Ge, W.Y., Jin, X.Q., Lin, W.J., Guo, W.H. and Yin, D.C., 2022. Antibacterial activity of lysozyme-loaded cream against MRSA and promotion of scalded wound healing. International Journal of Pharmaceutics, 627, p.122200.
Abstract. Staphylococcus aureus (S. aureus) infection, especially its drug-resistant bacterial infection, is a great challenge often faced by clinicians and patients, and it is also one of the most important threats to public health. Finding a safe and effective antibacterial agent is of great significance for the prevention and treatment of S. aureus infection. Lysozyme is known to have antibacterial effects against Gram-positive bacteria including S. aureus. Here, high-quality lysozyme with a purity of more than 99% and an activity of more than 60, 000 U/mg was prepared from egg white, which showed excellent antibacterial activity against three strains of S. aureus, especially against MRSA. Furthermore, an antibacterial cream loaded with lysozyme was prepared and tested in scald wound healing. The lysozyme-loaded cream exhibited the effect of preventing wound infection and promoting wound healing on scalds, and no toxicity was found in animal organs. Overall, lysozyme showed great application potential in the prevention and treatment of infections caused by S. aureus and scalded wound healing. The most remarkable discovery in this work is the unexpectedly powerful inhibitory effect of lysozyme on the drug-resistant bacterial, especially MRSA, which is usually very difficult to deal with using normal antibacterial drugs.
(4) Golac-Guzina, N., Novaković, Z., Sarajlić, Z., Šukalo, A., Džananović, J., Glamočlija, U., Kapo, B., Čordalija, V. and Mehić, M., 2019. Comparative study of the efficacy of the lysozyme, benzydamine and chlorhexidine oral spray in the treatment of acute tonsillopharyngitis-results of a pilot study. Acta Medica Academica, 48(2), pp.140-146.
Abstract. Objective. Lysozyme is a natural antimicrobial and immunomodullatory enzyme, which is produced as a host response to infectious agents. The objective of this study was to compare the efficacy and safety of lysozyme-based versus benzydamine and chlorhexidine-based oral spray in patients with an acute tonsillopharyngitis associaated with a common cold. Patients and Methods. A prospective two-arm pilot study (lysozyme/cetylpyridinium/lidocaine spray versus: benzydamine spray—arm 1; chlorhexidine/lidocaine spray—arm 2) was conducted in the primary health care unit. Efficacy was evaluated by the patient’s self-assessment of pain, difficulty in swallowing and the throat swelling, by using the visual analog scale (VAS) at baseline and three follow-up visits. Safety was evaluated by the assessment of the frequency and severity of adverse effects. Results. Lysozyme-based spray reduced pain faster than benzydamine-based spray and slower than chlorhexidine-based spray. Lysozyme-based and chlorhexidine-based sprays similarly reduced difficulty in swallowing, but were faster than benzydamine-based spray. Similar effects on the reduction of throat swelling were seen in all treated groups. All tested products showed proper safety and were well tolerated, with no serious adverse events reported. Conclusions. The lysozyme-based oral spray was shown to be effective and safe in the reduction of pain, difficulty in swallowing and throat swelling in patients with acute tonsillopharyngitis associated with a common cold. Lysozyme-based oral spray (containing natural compound with advantages of influencing immune system and preventing recurrences) had similar activity to benzydamine and chlorhexidine-based oral antiseptic sprays.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about E967 Review Consensus 10 by FRanier (9976 pt) | 2023-Apr-27 15:07 |
| Read the full Tiiip | (Send your comment) |
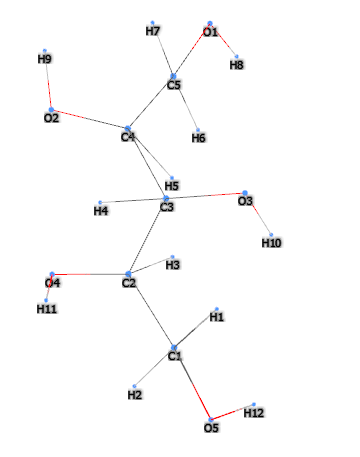
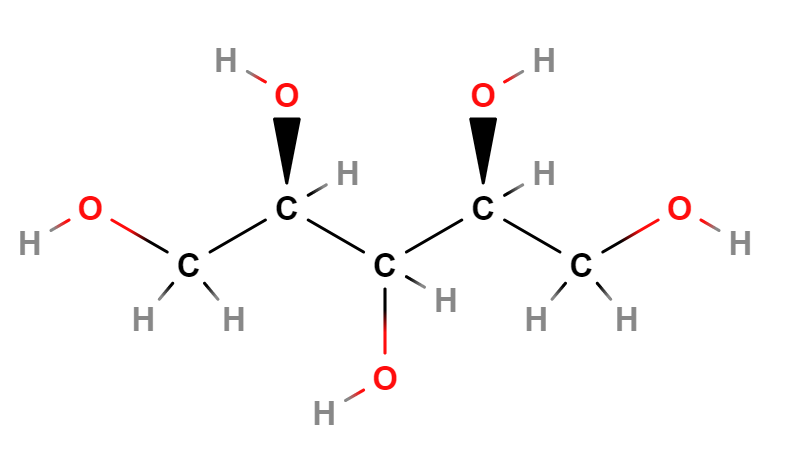
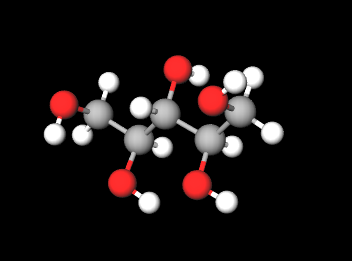
E967 (Xylitol) is a sugar alcohol with five carbon atoms and ethanol and water are used for its industrial extraction. It is produced naturally by mammalian metabolism.
It appears as a white crystalline powder.

What it is used for and where
Food
It is an artificial sweetener of vegetable origin that replaces sugar, labeled with the number E967 in the list of food additives, it is called "the sugar of wood" as it is made from some types of trees and corn. It is produced by the hydrogenation of Xylose in conditions of high temperature (80-140 ° C) and high pressure (up to 50 atm) (1).
It belongs to polyalcohols that have the characteristic of contrasting with acids that affect tooth enamel. This is why it is used in chewing gum and in the food and pharmaceutical fields.
After 1960, Xylitol has become a common ingredient in sugar-free baked goods, anticorrosive toothpaste and mouthwashes, oral care products and diabetic food.
Excessive intake may cause laxative effects.
Cosmetics
Anti-sebum. Controls and reduces emissions from the sebaceous glands, which are responsible for greasy, enlarged pores in the skin, where it occurs, particularly on the forehead, cheeks, nose and hair. Adjuvant in the treatment of acne.
Deodorant agent. When substances that give off an unpleasant odour are included in cosmetic formulations (typical examples are methyl mercaptan and hydrogen sulphide derived from garlic), deodorants attenuate or eliminate the unpleasant exhalation.
Flavoring agent. The purpose of this ingredient is to modify the solution to impart a certain flavour. Natural flavouring extracts are rather expensive, so the cosmetic and pharmaceutical industries resort to synthesised substances that have sensory characteristics mostly similar to natural flavourings or are naturally equivalent. This ingredient is isolated through chemical processes or is synthesised from chemicals. It is also referred to as Aroma.
Humectant. Hygroscopic compound used to minimise water loss in the skin and to prevent it from drying out by facilitating faster and greater absorption of water into the stratum corneum of the epidermis. The epidermis is the most superficial of the three layers that make up human skin (epidermis, dermis and hypodermis) and is the layer that maintains hydration in all three layers. In turn, the epidermis is composed of five layers: horny, the most superficial, granular, spinous, shiny, and basal. Humectants have the ability to retain the water they attract from the air in the stratum corneum and have the function of moisturising the skin. They are best used before emollients, which are oil-based.
Skin conditioning agent - Humectant. Humectants are hygroscopic substances used to minimise water loss in the skin and to prevent it from drying out by facilitating faster and greater absorption of water into the stratum corneum of the epidermis. The epidermis is the most superficial of the three layers that make up the human skin (epidermis, dermis and hypodermis) and is the layer that maintains hydration in all three layers. In turn, the epidermis is composed of five layers: corneum, the most superficial, lucidum, granulosum, spinosum and basale. Humectants have the ability to retain in the stratum corneum the water they attract from the air and have the function of moisturising the skin. It is better to use them before emollients that are oil-based.
Medical
It is used in the prevention of acute otitis media, respiratory diseases, parenteral nutrition, atopic dermatitis, wound repair, gastrointestinal infections, osteoporosis, anti-aging and inflammatory processes (2) and in food for diabetics.
The most relevant studies on this ingredient have been selected with a summary of their contents:
Xylitol studies
Typical commercial product characteristics Xylitol
| Appearance | White powder |
| Boiling Point | 494.5±40.0°C at 760 mmHg |
| Melting Point | 94-97°C(lit.) |
| Flash Point | 261.9±21.9°C |
| Loss on Drying | 5% |
| Sulphated Ash | 5% |
| Conductivity ash | 0.007 |
| Particle size > 2.4mm | 0 |
| Heavy Metal | 5ppm |
| As | 2ppm |
| Lead | <0.3 |
| Nickel | <1 |
| Total Plate | 1000/g Max |
| Yeast & Mold | 100/g Max |
| pH 10% w/v | 5.5 |
| Other polyols HPLC | <0.4 |
| Reducing sugars | 0.03 |
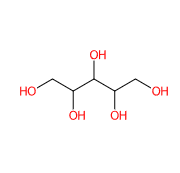 |  |
 |  |
Synonyms:
References_________________________________________________________________________
(1) Granström TB, Izumori K, Leisola M. A rare sugar xylitol. Part II: biotechnological production and future applications of xylitol. Appl Microbiol Biotechnol. 2007
Wisniak J, Hershkowitz M, Leibowitz R, Stein S. Hydrogenation of xylose to xylitol. Ind Eng Chem Res. 1974
(2) Da Silva, S.S. and Chandel, A.K. eds., 2012. D-xylitol: fermentative production, application and commercialization. Springer Science & Business Media.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about E951 Review Consensus 10 by FRanier (9976 pt) | 2023-Apr-26 19:03 |
| Read the full Tiiip | (Send your comment) |
E951 (Aspartame) (L-aspartyl-L-phenylalanine methyl ester) is a chemical compound formed from aspartic acid and phenylalanine.
Industrially it occurs as a fine white powder.

What it is used for and where
Food
The history of aspartame dates back to 1965 when the American chemist and researcher James Schlatter, in an attempt to create a drug against gastric ulcer, chemically derived aspartame in an intermediate step of creating a tetrapeptide of the peptide hormones gastrin.
It is one of the most common sweeteners and on which have been focused countless scientific researches to establish the risks connected to the consumption of this product which is included in many food products: diet drinks, chewing gums, yogurt, desserts, vitamins, medicines. It is recommended to people with diabetes as a substitute of sugar. It has about 200 times the sweetening power of sugar.
In the European Union it is labeled as E951 in the list of european food additives as a sweetener and it is considered by Efsa (European Food Safety Authority), safe for human health.
The acceptable daily intake of aspartame recommended by the FDA is 50 mg/kg per day in the United States and 40 mg/kg per day in the European Union (EFSA).
Researchers from the Schiller Institute for Integrated Science and Society, Boston, based on the 1997 toxicological studies of the Ramazzini Institute (an independent non-profit research laboratory based in Bologna) carried out on laboratory animals, concluded that aspartame is a chemical carcinogen in rodents even at the dose of 100 mg/kg body weight, a relatively low dose exposure dangerously close to the current permissible daily intake (ADI) levels in the USA and the European Union (1).
High-intensity sweeteners are considered potential organic contaminants due to their widespread use in food, drugs and healthcare products. They are introduced into the environment by different pathways and find their way into water (2).
For more information:
Typical optimal characteristics of the commercial product Aspartame
| Appearance | Powder fine white |
| Density | 1.3±0.1 g/cm3 |
| Alpha | 15.5 º (c=4, 15N formic acid) |
| Melting Point | 242-248 °C |
| Boiling Point | 535.8±50.0 °C at 760 mmHg |
| Flash Point | 277.8±30.1 °C |
| 14.5 ° (C=4, 15mol/L Formic Acid) | |
| Vapour Pressure | 0.0±1.5 mmHg at 25°C |
| Storage | 2-8°C |
| Solubility | Sparingly soluble or slightly soluble in water and in ethanol (96 per cent), practically insoluble in hexane and in methylene chloride. |
| Pka | pKa 3.19±0.01 (H2O t=25.0 I=0.100(NaCl))(Approximate);7.87±0.02(H2O t=25.0 I=0.100(NaCl))(Approximate) |
 |  |
 |  |
 |  |
Synonyms:
References_____________________________________________________________________
(1) Landrigan PJ, Straif K. Aspartame and cancer - new evidence for causation. Environ Health. 2021 Apr 12;20(1):42. doi: 10.1186/s12940-021-00725-y.
(2) Arbeláez P, Borrull F, Pocurull E, Marcé RM. Determination of high-intensity sweeteners in river water and wastewater by solid-phase extraction and liquid chromatography-tandem mass spectrometry. J Chromatogr A. 2015 May 8;1393:106-14. doi: 10.1016/j.chroma.2015.03.035.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about E418 Review Consensus 9 by FRanier (9976 pt) | 2023-Apr-14 15:59 |
| Read the full Tiiip | (Send your comment) |
E418 (Gellan gum) is a heteropolysaccharide, an anionic exocellular polysaccharide produced by Sphingomonas elodea in a carbohydrate fermentation process in which glucose, glucuronic acid, rhamnose and esterified acetic acid are also separately involved.
It appears in the form of a white powder.

Gellan is a natural hydrocolloid polysaccharide with thickening properties from which gellan gum is obtained, used in medical, food and in the restoration of ancient works of art, especially books.
What it is used for and where
Food
In the food industry, it is an additive listed as E418 in the European food additives list that is used as a thickener, stabiliser and emulsifier.
Cosmetics
Emulsion stabilizer. Emulsions are thermodynamically unstable. Emulsion stabilisers improve the formation and stability of single and double emulsions. It should be noted that in the structure-function relationship, molar mass plays an important role.
Viscosity Enhancing Agent - aqueous. Since viscosity is important for increasing the chemical and physical stability of the product, Viscosity Enhancing Agent acqueous is an important dosage factor in gels, suspensions, emulsions, solutions. Increasing viscosity makes formulations less sedimentary and more homogeneously thickened.
Medical
A 'true gel structure' of Gellan gum is formed by the aggregation of these associations and is mediated by monovalent and/or divalent cations (e.g. Ca 2+ ) (1). Gellan gum has already been used, made into mucoadhesive beads with Ketoprofen (2) (a non-steroidal anti-inflammatory drug), particles with ciprofloxacin for skin applications (3), or in biogas compositions for bone tissue regeneration (4).
Other uses
In restoration, it is used for oxide reduction, cleaning and de-acidification.
CAS 71010-52-1
References__________________________________________________________________
(1) De Silva DA, Poole-Warren LA, Martens PJ, Panhuis M. Mechanical characteristics of swollen gellan gum hydrogels. Appl Polym Sci. 2013;130(5):3374–3383. doi: 10.1002/app.39583.
(2) De Silva DA, Poole-Warren LA, Martens PJ, Panhuis M. Mechanical characteristics of swollen gellan gum hydrogels. Appl Polym Sci. 2013;130(5):3374–3383. doi: 10.1002/app.39583
(3) Novac O, Lisa G, Profire L, Tuchilus C, Popa MI Antibacterial quaternized gellan gum based particles for controlled release of ciprofloxacin with potential dermal applications. Mater Sci Eng C Mater Biol Appl. 2014 Feb 1; 35():291-9.
(4) Gantar A, da Silva LP, Oliveira JM, Marques AP, Correlo VM, Novak S, Reis RL Nanoparticulate bioactive-glass-reinforced gellan-gum hydrogels for bone-tissue engineering. Mater Sci Eng C Mater Biol Appl. 2014 Oct; 43():27-36.
Giavasis I, Harvey LM, McNeil B. Gellan gum. Crit Rev Biotechnol. 2000;20(3):177-211. doi: 10.1080/07388550008984169.
Abstract. For decades microbial exopolysaccharides have been invaluable ingredients in the food industry, as well as having many attractive pharmaceutical and chemical applications. Gellan gum is a comparatively new gum elaborated by the Gram-negative bacterium Sphingomonas paucimobilis. Although its physico-chemical properties have been well characterized, the ecology and physiology of Sphingomonas, and the factors influencing the fermentation process for production of this gum have received much less attention. This review focuses on the metabolism and the enzymic activity of this bacterium, as well as the factors that influence gellan production, including process temperature, pH, stirring rate, oxygen transfer, and composition of the production medium. Potential strategies for improving the production process are discussed in the context of processes for the production of other microbial biopolymers, particularly exopolysaccharides. In addition, the importance and potential utility of gellan lyases in modification of gellan and in other applications is critically evaluated.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
| "Descrizione" about E375 Review Consensus 10 by FRanier (9976 pt) | 2023-Apr-13 18:40 |
| Read the full Tiiip | (Send your comment) |
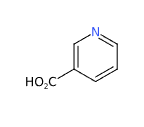
E375 or Nicotinic acid or Niacin (pyridine-3-formic acid) an organic vitamin, is also known as Vitamin B3 , (a generic name that also includes vitamin PP, niacinamide, nicotinamide and derivatives) is one of the 8 water-soluble B-complex vitamins.
It is found in :
Industrially it appears in the form of a white powder.

What it is used for and where
Medical
It is important for the conversion of proteins, carbohydrates and fats into energy.
It can reduce harmful LDL-cholesterol (which thickens and clogs the arteries), improve HDL-cholesterol values and thus reduce the risks for the cardiovascular system (1).
It also has positive indications for diabetes mellitus by bringing glycaemic values back to normal (2).
Food
Its deficiency can develop pellagra-like symptoms and is linked to a state of malnutrition. Ingredient included in the list of European food additives as E375 under the name nicotinic acid, acting as a stabiliser.
It is used in dairy products, in pasta making and in the production of maize flour.
Animal feed
Feed additive, which can improve milk production, poultry and meat quality.
Cosmetics
Skin conditioning agent - Mixed. This ingredient is responsible for modifying the condition of the skin when it is damaged or dry by reducing flaking and restoring elasticity.
Antistatic agent. Static electricity build-up has a direct influence on products and causes electrostatic adsorption. The antistatic ingredient reduces static build-up and surface resistivity on the surface of the skin and hair.
The most relevant studies on this vitamin have been selected with a summary of the contents:
 | 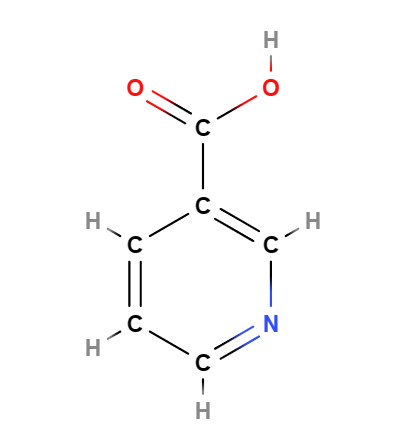 |
 | 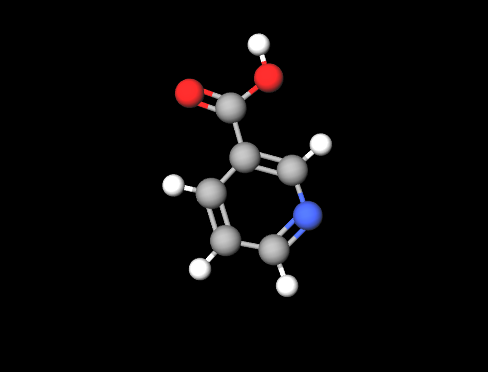 |
Synonyms:
References____________________________________________________________________
(1) Landray MJ, Haynes R, Armitage J. Niacin for reduction of cardiovascular risk. N Engl J Med. 2014 Nov 13;371(20):1943-4. doi: 10.1056/NEJMc1411240.
Niacin for reduction of cardiovascular risk.
Mayer L.
N Engl J Med. 2014 Nov 13;371(20):1943. doi: 10.1056/NEJMc1411240#SA6.
Niacin for reduction of cardiovascular risk.
Santos-Gallego CG, Badimon J.
N Engl J Med. 2014 Nov 13;371(20):1943. doi: 10.1056/NEJMc1411240#SA5.
Niacin for reduction of cardiovascular risk.
van den Oever IA, Nurmohamed MT, Lems WF.
N Engl J Med. 2014 Nov 13;371(20):1942. doi: 10.1056/NEJMc1411240#SA4.
(2) Ding Y, Li Y, Wen A. Effect of niacin on lipids and glucose in patients with type 2 diabetes: A meta-analysis of randomized, controlled clinical trials. Clin Nutr. 2014 Sep 28. pii: S0261-5614(14)00247-7. doi: 10.1016/j.clnu.2014.09.019.
Wu L, Parhofer KG. Diabetic dyslipidemia. Metabolism. 2014 Aug 29. pii: S0026-0495(14)00258-3. doi: 10.1016/j.metabol.2014.08.010.
| Sign up to vote this object, vote his reviews and to contribute to Tiiips.EvaluateClose | (0 comments) |
Read other Tiiips in __Italiano (172)
